As filed with the Securities and Exchange Commission on February 6, 2015
Registration No. 333-199477

UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Amendment No. 2
To
Form S-1
REGISTRATION STATEMENT
Under The Securities Act of 1933
CollabRx, Inc.
(Exact name of Registrant as specified in its Charter)
|
Delaware
|
68-0370244
|
|
|
(State or Other Jurisdiction of Incorporation or Organization)
|
(I.R.S. Employer Identification No.)
|
|
|
44 Montgomery Street, Suite 800
|
||
|
San Francisco, California
|
94104
|
|
|
(Address of Principal Executive Offices)
|
(Zip Code)
|
|
|
Primary Standard Classification Code Number:
|
7374
|
Registrant’s telephone number, including area code: (415) 248-5350
Thomas R. Mika
President and Chief Executive Officer
44 Montgomery Street, Suite 800
San Francisco, CA 94104
(415) 248-5350
(Name, address, including zip code, and telephone number, including area code, of agent for service)
Copies to:
|
William Davison, Esq.
|
Gregory Sichenzia, Esq.
|
|
|
Goodwin Procter LLP
|
Marcelle Balcombe, Esq.
|
|
|
135 Commonwealth Drive
|
Jeff Cahlon, Esq.
|
|
|
Menlo Park, California 94025
|
Sichenzia Ross Friedman Ference LLP
|
|
|
Tel: (650) 752-3114
|
61 Broadway
|
|
|
New York, NY 10006
|
||
|
Tel: (212) 930-970
|
Approximate date of commencement of proposed sale to the public: As soon as practicable after the effective date of this registration statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, as amended, check the following box. ☑
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer or a smaller reporting company. See definitions of “large accelerated filer”, “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
|
Large accelerated filer ☐
|
Accelerated filer ☐
|
|
Non-accelerated filer ☐ (Do not check if a smaller reporting company)
|
Smaller reporting company ☑
|
The registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933 or until this registration statement shall become effective on such date as the Commission, acting pursuant to said Section 8(a), may determine.
2
The information in this preliminary prospectus is not complete and may be changed. These securities may not be sold until the registration statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell nor does it seek an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
|
PRELIMINARY PROSPECTUS
|
SUBJECT TO COMPLETION
|
DATED FEBRUARY 6, 2015
|
Up to $3.0 Million in Shares of Common Stock and Warrants to Purchase Shares of Common Stock

We are offering up to shares of common stock, together with warrants to purchase additional shares of common stock at a purchase price of $ (and the shares issuable from time to time upon exercise of the warrants) pursuant to this prospectus. The shares and warrants will be separately issued, but the shares and warrants will be issued and sold to purchasers in equal proportion. Each warrant will have an exercise price of $ per share, will be exercisable upon issuance and will expire five years from the date of issuance.
Our common stock is listed on the NASDAQ Capital Market under the symbol “CLRX.” The last reported sale price of our common stock on The NASDAQ Capital Market on February 5, 2015 was $1.52 per share. There is no established public trading market for the warrants, and we do not expect a market to develop. In addition, we do not intend to apply for a listing of the warrants on any national securities exchange.
Investing in the offered securities involves a high degree of risk. See “Risk Factors” on page 19 to read about factors you should consider before investing in our securities.
Neither the Securities and Exchange Commission nor any other regulatory body has approved or disapproved of these securities or passed upon the accuracy or adequacy of this prospectus. Any representation to the contrary is a criminal offense.
|
Per Share
and
Warrant
|
Total
|
|||||||
|
Public Offering Price
|
$
|
$
|
||||||
|
Underwriting Discounts and Commissions(1)
|
$
|
$
|
||||||
|
Proceeds to us, before expenses
|
$
|
$
|
||||||
(1) Does not include a non-accountable expense allowance equal to 1% of the gross proceeds of this offering payable to the underwriter. See “Underwriting” beginning on page 106 of this prospectus for a description of the compensation payable to the underwriter.
3
The underwriter may also purchase from us up to an additional shares of common stock and warrants to purchase additional shares of common stock at the public offering price, less the underwriting discount, within 45 days from the date of this prospectus to cover overallotments, if any.
The underwriter expects to deliver the shares and warrants against payment therefore on or about February , 2015.
Aegis Capital Corp
4
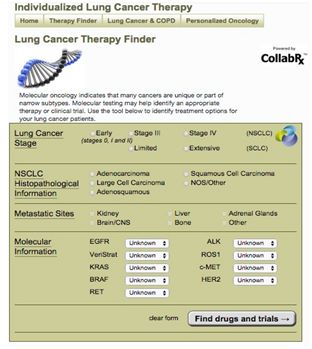
 This input page from CollabRx’ Lung Cancer Therapy Finder prompts physicians to enter the relevant data regarding their patient’s tumor. Included are those molecular tests that CollabRx’ Lung Cancer Advisory Board believes are the most relevant (based on clinical evidence) and which, when combined with other information about stage, histopathology, metastatic sites and in some cases prior treatments, should be considered in guiding treatment planning. The output of this interactive app is a description of the importance of each relevant test result, along with fully annotated lists of drugs, clinical trials and citations to the published evidence that supports the relationship of the biomarkers to these therapies.
|
CancerRx - an IOS application designed to help oncologists and pathologists navigate the complex landscape of oncology therapeutic options. CancerRx was formally introduced to physicians at the 2014 American Society of Clinical Oncology (ASCO) Annual Meeting that took place May 30-June 3, 2014 in Chicago.
Highlights of the ASCO launch include:
● More than 9,600 downloads (5/30-6/11)
● Averaging 1,677 weekly unique users
● Target users include oncologists and other health care
practitioners involved in cancer care (approximately 40,000 potential users)
● A 5-star rating on the Apple App Store
 |
5
 |
|
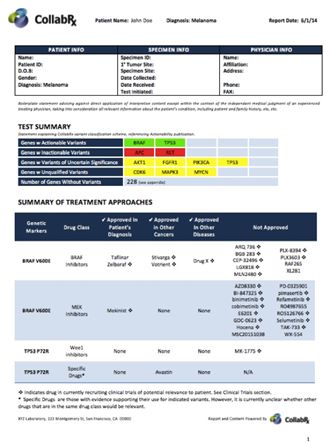
CollabRx’s Genetic Variant Annotation™ Service (GVA™) supports clinical diagnostic laboratories that perform tumor genomic testing on cancer tumors to uncover genetic alterations that may lead to novel therapeutic approaches for some cancer patients. Offered as Software-as-a-Service (SAAS), diagnostic labs provide the digital output of DNA sequencing and other analytical devices to CollabRx for the identification of “actionable” biomarkers. The GVA provides information back to the laboratory regarding what is known about the actionable biomarker and, as relevant, additional information about related drugs and clinical trials – all in a fully automated system.
|
|
Page
|
|
|
8
|
|
|
19
|
|
|
36
|
|
|
38
|
|
|
39
|
|
|
39
|
|
|
40
|
|
|
42
|
|
|
44
|
|
|
67
|
|
|
83
|
|
|
88
|
|
|
97
|
|
|
98
|
|
|
99
|
|
|
103
|
|
|
105
|
|
|
113
|
|
|
113
|
|
|
113
|
|
|
113
|
|
|
115
|
We have not authorized anyone to provide you with any information or to make any representation, other than those contained in this prospectus or any free writing prospectus we have prepared. We take no responsibility for, and provide no assurance as to the reliability of, any other information that others may give you. This prospectus is an offer to sell only the securities offered hereby, but only in circumstances and in jurisdictions where it is lawful to so do. The information contained in this prospectus is accurate only as of its date, regardless of the time of delivery of this prospectus or of any sale of our securities.
Neither we nor the underwriter has done anything that would permit this offering or possession or distribution of this prospectus in any jurisdiction or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than the United States. You are required to inform yourself about, and to observe any restrictions relating to, this offering and the distribution of this prospectus.
This summary highlights selected information contained elsewhere in this prospectus and does not contain all of the information that you should consider in making your investment decision. Before investing in our securities, you should carefully read this entire prospectus, including our financial statements and the related notes included elsewhere in this prospectus. You should also consider, among other things, the matters described under “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” in each case appearing elsewhere in this prospectus. Unless otherwise stated, all references to “us,” “our,” “CollabRx,” “we,” the “Company” and similar designations refer to CollabRx, Inc.
Overview
CollabRx, Inc. develops and markets medical information and clinical decision support products and services intended to set a standard for the clinical interpretation of genomics-based, precision medicine. Building on a comprehensive and specialized knowledge base, software systems that facilitate dynamic updating and automated reporting, and a unique network of over 75 independent expert clinical advisors, we have developed a suite of unique and differentiated information products and services. These products include a scalable system for providing high-value information on biomarkers and related therapies for inclusion in reports prepared by diagnostic labs reporting on the results from their multi-biomarker genomic-based tests. In addition, we have developed decision-support tools for physicians and patients to assist with treatment planning for advanced cancer, offered over the Internet in both web-based and mobile apps. We believe that both product sets are highly synergistic, and allow opportunities for enhancement, cross-fertilization and adoption that go beyond what each would offer individually.
Our overall vision is that we are at the dawn of an era of explosive growth of data and information generated at the molecular level that must be interpreted and contextualized into knowledge before it can be used effectively by either physicians or patients. We regard this knowledge as being the most valuable portion of the molecular diagnostic process and we believe that all sectors of the healthcare industry, including providers, insurers, drug developers and patients, are potential users of this knowledge. We aim to deliver our proprietary interpretive content as quickly as possible and in as many usable forms as possible via the Internet.
We are just entering our commercialization phase. The systems and approach that we have developed can be applied to many disease states, but we have chosen to focus initially on cancer, an area of tremendous need and potential. We believe that our approach is unique, disruptive in the market and well timed, offering the potential for high growth, profitability and above average returns for investors.
Products and Services
We support oncologists and pathologists in their efforts to plan treatment approaches for their advanced cancer patients. We do so through three major information products, all of which are based on a proprietary knowledgebase that is informed by external data sources, our in-house experts, and a large network of independent clinical advisors. We attempt to gather together much of the data and evidence that is relevant to different therapeutic approaches in the context of the presence of specific genetic biomarkers, relating those to the relevant drugs and clinical trials that may help a physician plan a treatment approach for a patient. Similar to an electronic library, we focus on keeping the information comprehensive, accurate, current and easy to access. Currently, our information products include the following:
|
Product
|
Users
|
Description
|
Business Model
|
|
Genetic Variant Annotation Service™
(GVA™)
|
Pathologists and Laboratory Medical Directors via cloud-based servers
|
Automated clinical interpretation of tumor genetic alterations (mutation and copy number variation)
|
Laboratories pay $75-$150 per test event or purchase annual subscription
|
|
Therapy Finders™
for Melanoma, Colorectal and Lung Cancer and Metastatic Breast Cancer
|
Oncology professionals at the point-of-care
|
Web-based expert systems for clinical decision support
|
Advertising and sponsor-ship sharing with on-line media partner MedPage Today
|
|
CancerRx
|
Oncology professionals at the point-of care
|
Mobile app with reference tools, social media, and expert systems
|
Advertising and sponsor-ship sharing with media partner MedPage Today
|
Since launching our Genetic Variant Annotation Service (GVA) in August 2013, several prominent diagnostic laboratories, comprehensive cancer centers and life science companies have become customers of and/or partners in this product, including: Life Technologies, Inc. (now Thermo-Fisher, Inc., Carlsbad, California), Affymetrix, Inc. (Santa Clara, California), OncoDNA, SA (Brussels, Belgium), Sengenics, Pte., Ltd. (Singapore), Cynvenio Biosystems, Inc. (Westlake Village, California), Quest Diagnostics, Inc. (Madison, New Jersey), CellNetix Pathology and Laboratories (Seattle, Washington), Genoptix (a Novartis company, Carlsbad, California), The Jackson Laboratory for Genomic Medicine (Bar Harbor, Maine), The University of Chicago Medical (Chicago, Illinois), Stanford Hospital Molecular Laboratory (Palo Alto, CA) and The Ohio State University Medical Center (Columbus, Ohio).
Our Therapy Finders™ and CancerRx are made available free to physicians and patients via our on-line media partner, MedPage Today, the professional property of Everyday Health, Inc.
The Cancer Market and Genomic Testing
Cancer is a worldwide health concern. In 2002, cancer eclipsed heart disease as the number one cause of death in the U.S. for individuals under the age of 85, according to the National Cancer Institute. In the U.S., nearly 12 million individuals are living with a cancer diagnosis, approximately 7 million individuals are being treated for cancer, approximately 1.5 million new cases are diagnosed each year, over 600,000 people die from cancer each year (6 million deaths worldwide), and nearly 1 in 3 females and 1 in 2 males will be diagnosed with cancer in their lifetime, according to the American Cancer Society. Age is the number one risk factor for the development of cancer. Nearly 80% of cancers are diagnosed in individuals age 55 years and older, which is the fastest growing segment of the U.S. population. JPMorgan recently estimated that the total market opportunity for testing in cancer is estimated at $10B in 2014 in the U.S. alone, growing to $25B by 2018, but this opportunity has only been partially tapped.
Cancer treatment and research has been experiencing an unprecedented explosion of knowledge in the past 5-10 years. There are over 500 new cancer therapies in pre-clinical development, thousands of diagnostic labs (private and hospital based), more than 10,000 cancer-related clinical trials are currently ongoing, and over 100,000 cancer-related papers are published in the scientific literature annually, according to PubMed and ClinicalTrials.gov. The knowledge explosion is associated with two interrelated key drivers: (i) cancer is fundamentally a disease of the genome, (i.e., genetic mutations cause cancer), and (ii) the costs and time to sequence genes (and discover mutations that can be targeted for therapy) have been falling at an increasing rate, driven principally by new sequencing technologies commonly referred to as “Next Generation Sequencing” or “NGS”. The concept of a “$1,000 genome” has broad psychological implications since it represents the price point at which leading experts believe that every cancer genome will be sequenced.
Large-scale genetic sequencing studies in tumors are rapidly uncovering mutations in genes that can be selectively targeted by pharmaceutical and biotechnology companies. There were fewer than 10 cancer genes in 2008 in which genetic aberrations were strongly associated with treatment implications. By comparison, there is broad consensus that there are currently approximately 50 such genes based on recent studies, a number that is expected to double in the next year even as additional mutations are being discovered in genes already associated with cancer. Currently there are approximately over 95 biomarkers representing thousands of mutations in aggregate that are associated with at least some level of clinical actionability. This number is expected to increase rapidly as new discoveries are made.
Competitive Strengths
CollabRx is differentiated and unique as an information company. We are not a diagnostic lab offering a particular test or series of tests in cancer diagnostics. Instead, we have focused exclusively on the information, analysis and interpretation-based steps in the diagnostic workflow, developing and refining the increasingly complex task of delineating the relationship between known or studied biomarkers in cancer with the therapeutic strategies that the published evidence supports. With our web-based and mobile apps, we provide a means for physicians to access our knowledgebase easily at the point-of-care. For laboratories, we provide a credible, third party resource for the dynamic information and analysis that is needed to interpret the results of genetic tests.
Our conviction is that the interpretation and reporting of genomic-based test results will become the key differentiator in the market, as opposed to the design and performance of the test itself, given the rapid commoditization of NGS data generation and inherent lack of intellectual property in the sequencing steps. Ultimately, we believe physicians will judge the quality of a diagnostic test based on the quality of the report, and how well it supports the treatment decision process. We are platform agnostic, independent, adaptable and unregulated. We believe that diagnostic companies, medical centers, hospital labs and other community-based labs interested in developing a genomics testing capability will confront the challenges associated with developing and maintaining a clinically-oriented, evidence-based biomarker reference database, and increasingly will realize that it is better to “buy” than to “build.”
In addition, we believe that it is important to address physician needs for information directly, rather than solely via diagnostic laboratories. For this reason we have continued the development of our web-based and mobile applications, addressing oncologists and pathologists at work and at home, providing a resource for both education and for decision-support.
Our ability to compete in these markets and our ability to serve the needs of physicians treating advanced cancer patients rests on a set of principles and ideas that are potentially very disruptive to the markets that we serve and which offer an opportunity for extraordinary growth and profitability. We believe that the following attributes of CollabRx provide a sustainable competitive advantage:
|
·
|
Our proprietary knowledgebase is focused on actionable information for physicians – CollabRx medical and scientific content is organized in a knowledgebase that expresses the relationship between genetic profiles, other aspects of the medical record (e.g., stage, prior treatments), and therapy considerations including molecular diagnostics, medical tests, clinical trials, drugs, biologics, and other information relevant for treatment planning. Our focus is, and always has been on providing actionable information that physicians can use to plan treatment strategies for their advanced cancer patients and identifying the evidence in the public domain that justifies the therapy options presented. |
|
·
|
Our automated software platform is scalable and capable of handling high test volumes and fast turn-around times – The CollabRx “Semantic Integration Platform” or SIP brings together methods track important changes in molecular oncology from numerous sources, including the published literature and many of the centralized publicly available databases utilized by biomedical and translational clinician/scientists. Our SIP provides CollabRx with a scalable, interactive service that can handle large test volumes and still maintain fast turn-around times for our customers. In this respect it is unmatched in our field. |
|
·
|
Our large network of independent expert clinical advisors – Over 75 independent, uncompensated expert advisors, organized by both tissue-specific editorial boards and pan-cancer or biomarker-centric boards, provides a unique, unbiased mechanism to inform and prioritize treatment strategies based on evidence. |
|
·
|
Our first-mover advantage and independence - We believe that CollabRx is the first company to have focused exclusively on the information-based, value-added steps of the diagnostic testing workflow in the context of providing clinical grade interpretation of multi-gene testing in cancer, separate from the processing of tissue samples in a laboratory environment. |
Growth Strategy
Our strategic vision is to become a leading source of high value, independently reviewed, actionable information on molecular medicine that supports decision-making by physicians and patients, embodied in tools and services whose performance or use allow us to accumulate data that is valuable to paying segments of the health care ecosystem. We intend to build our information franchise first in oncology, and then expand to other genomics-based disease areas that are amenable to our approach of automated, scalable, and expert-vetted content creation and distribution.
Our growth strategy includes the following key elements:
|
·
|
Marketing of our Genetic Variant Annotation™ Service into additional segments within the clinical diagnostic laboratory market. |
|
·
|
Forming strategic partnerships with life science companies to expand GVA biomarker coverage and utility and to engage with them in cooperative marketing efforts. |
|
·
|
Forming partnerships with companies in the diagnostic testing workflow to enhance the GVA and to expand our customer base. |
|
·
|
Supporting the advertising and sponsorship sales activities of Everyday Health, Inc. in connection with the current and future Therapy Finders and CancerRx mobile apps. |
Risks Associated with Our Business
An investment in our common stock involves a high degree of risk. Any of the factors set forth under “Risk Factors” may limit our ability to successfully execute our business strategy. You should carefully consider all of the information set forth in this prospectus and, in particular, should evaluate the specific factors set forth under “Risk Factors” in deciding whether to invest in our securities. Some of the principal risks we face are:
|
·
|
we may not be able to generate sufficient cash flow or raise capital on acceptable terms to meet our needs; |
|
·
|
we may not be able to successfully implement our growth on a timely basis or at all; |
|
·
|
we may lose key members of our senior management team; |
|
·
|
our products may be alleged to be faulty or fail to comply with government regulation; |
|
·
|
we may lose a significant customer; and |
|
·
|
our business development and marketing programs may prove insufficient or ineffective. |
Company Background
CollabRx (f/k/a Tegal) was formed in December 1989 to acquire the operations of the former Tegal Corporation, or Tegal, a division of Motorola, Inc. Until recently, we designed, manufactured, marketed and serviced specialized systems used primarily in the production of semiconductors and micro-electrical mechanical devices, including integrated circuits, memory devices, sensors, accelerometers and power devices. Beginning in late 2009, we experienced a sharp decline in revenues resulting from the collapse of the semiconductor capital equipment market and the global financial crisis. In a series of transactions from 2010 to 2012, we sold the majority of our operating assets and intellectual property portfolio. During the same time period, our board of directors evaluated a number of strategic alternatives, which included our continued operation as a stand-alone business with a different business plan, a merger with or into another company, a sale of our remaining assets, and our liquidation or dissolution. We investigated opportunities within and outside the semiconductor capital equipment industry and evaluated a number of transactions involving other diversified technology-based companies. Throughout this process, we developed and refined our criteria for a business combination, with an eventual focus on the healthcare industry, and specifically information technology and services within the healthcare industry.
Company and Other Information
We were formed in December 1989 to acquire the operations of the former Tegal Corporation, a division of Motorola, Inc. Our predecessor company was founded in 1972 and acquired by Motorola in 1978. We completed our initial public offering in October 1995. On July 12, 2012, we completed the acquisition of a private company called CollabRx, Inc., herein referred to as the Merger, pursuant to an Agreement and Plan of Merger dated as of June 29, 2012. As a result of the Merger, CollabRx, Inc. became a wholly-owned subsidiary of ours. In consideration for 100% of the stock of CollabRx, Inc., we issued an aggregate of 236,433 shares of common stock, representing approximately 14% of our total shares outstanding prior to the closing, to former CollabRx, Inc. stockholders. We subsequently changed our name to CollabRx, Inc.
Our principal executive office is located at 44 Montgomery Street, Suite 800, San Francisco, CA 94104, and our telephone number is (415) 248-5350. Our website address is www.collabrx.com. We do not incorporate the information on or accessible through our website into this prospectus, and you should not consider any information on, or that can be accessed through, our website as part of this prospectus.
We own two U.S. federal trademark registrations and applications, and unregistered trademarks and servicemarks, including CollabRx, Inc.™, and Therapy Finders™. All other trademarks or trade names referred to in this prospectus are the property of their respective owners. Solely for convenience, the trademarks and trade names in this prospectus are referred to without the ® and ™ symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. We do not intend the use or display of other companies’ trademarks and trade names to imply a relationship with, or endorsement or sponsorship of us by, any other companies.
Recent Developments
Nasdaq Notices
On November 18, 2014, we were notified by Nasdaq that the bid price of the our common stock closed below the minimum $1.00 per share requirement for continued inclusion under Marketplace Rule 4310(c)(4). In accordance with Marketplace Rule 4310(c)(8)(D), we have180 calendar days, or until May 18, 2015, to regain compliance. If at anytime before May 18, 2015, the bid price of our common stock closes at $1.00 per share or more for a minimum of 10 consecutive business days, we will regain compliance with the Rule. If we do not regain compliance by May 18, 2015, an additional 180 days will be granted to regain compliance, so long as we meet The Nasdaq Capital Market initial listing criteria (except for the bid price requirement).
On November 20, 2014, we were notified by Nasdaq that the shareholders' equity balance reported on our last Quarterly Report filed with the Securities and Exchange Commission on November 14, 2014 fell below the $2,500,000 minimum requirement for continued listing under the Nasdaq Capital Market's Listing Rule 5550(b)(1). On January 20, 2015, we announced that we received a letter from the NASDAQ Listing Qualifications Staff indicating that, unless we timely request a hearing before the NASDAQ Listing Qualifications Panel, our securities would be delisted from The NASDAQ Capital Market due to our non-compliance with NASDAQ Listing Rule 5550(b)(1). We have requested a hearing before the Panel, at which hearing we will present our plan to evidence compliance with Rule 5550(b)(1), which requires us to maintain a minimum of $2.5 million in stockholders' equity. Our common stock will continue to trade on The NASDAQ Capital Market under the symbol "CLRX" pending completion of the hearing process and the expiration of any extension period granted by the Panel.
Proposed Transaction with Medytox Solutions, Inc.
On December 6, 2014, we entered into a non-binding letter of intent with Medytox Solutions, Inc. for a potential business combination between the companies. The business combination is subject to, among other things, due diligence, the execution of a definitive agreement, necessary board of director and stockholder approvals and other customary conditions. We cannot assure you that we will enter into a definitive agreement and, if such definitive agreement is entered into, that the contemplated business combination will be consummated.
Pursuant to the Letter of Intent, Medytox Solutions agreed to advance certain funding to CollabRx in contemplation of the business combination. On January 16, 2015, we entered into a Loan and Security Agreement with MedytoxSolutions, pursuant to which it is contemplated that Medytox Solutions will loan up to $2,395,644 to CollabRx. We intend to use the proceeds from the Loan and Security Agreement for working capital and general corporate purposes. Amounts borrowed by our company under the Loan and Security Agreement will accrue simple interest at the rate of fifteen percent (15.0%) per annum. As of February 4, 2015, we had borrowed $440,715 under the Loan and Security Agreement. The making of additional advances to our company under the Loan and Security Agreement is completely discretionary on the part of MedytoxSolutions. All amounts borrowed under the Loan and Security Agreement mature on December 31, 2015. Upon the occurrence of an event of default under the Loan and Security Agreement, all or a portion of the then outstanding principal and accrued interest under the Loan and Security Agreement is convertible, in the discretion of MedytoxSolutions, into shares of our common stock at a conversion price equal to the lower of (i) $0.85 or (ii) the average of the bid and ask prices of ourcommon stock on the trading day immediately prior to the date of conversion; provided, however, that the maximum number of shares issuable to Medytox Solutions is 14.9% of the number of shares of common stock then outstanding. We agreed to secure the payment and performance of our obligations under the Loan and SecurityAgreement by the grant of a security interest in all of our assets.
The Loan Agreement includes representations and warranties of the parties, covenants and agreements regarding the operation of our business while amounts are outstanding under the Loan and Security Agreement, and indemnification provisions in the event of a breach of a representation, warranty, covenant or agreement contained in the Loan and Security Agreement.
Also on January 16, 2015, we entered into an Agreement with MedytoxSolutions, pursuant to which we agreed that in the event we enter into a merger or other sale transaction involving at least thirty-five percent (35.0%) of our shares or assets with a party other than MedytoxSolutions, we will pay Medytox Solutions a $1,000,000 fee. Notwithstanding the foregoing, no fee will be payable to Medytox Solutions in the event (i) MedytoxSolutions has not provided funding to our company of at least $500,000 pursuant to the Loan and Security Agreement or (ii) MedytoxSolutions has not funded an advance requested by us under the Loan and Security Agreement, subject to certain exceptions.
THE OFFERING
|
Securities offered by us:
|
Up to shares of common stock and warrants to purchase additional shares of common stock
|
|
Common stock to be outstanding after this offering:
|
Shares ( if the warrants are exercised in full). If the underwriter exercises its option to purchase additional shares in full, the total number of shares of common stock outstanding immediately after this offering would be shares ( shares if the warrants are exercised in full).
|
|
Overallotment option to purchase additional shares and warrants:
|
We have granted the underwriter a 45-day option to purchase from us up to an additional shares of our common stock and warrants to purchase additional shares of common stock at the public offering price, less underwriting discounts and commissions.
|
|
Description of warrants:
|
The warrants will be separately transferable immediately upon issuance, but the warrants will be issued and sold to purchasers in equal proportion as shares. Each warrant will have an exercise price of $ per share, will be exercisable upon issuance and will expire five years from the date of issuance.
|
|
Use of proceeds by us:
|
We estimate that we will receive net proceeds from this offering of up to approximately $2.6 million based upon an assumed public offering price of $1.52 per share, which is the last reported sale price of our common stock on the NASDAQ Capital Market on February 5, 2015 (or up to approximately $3.0 million if the underwriter’s option to purchase additional shares and warrants in this offering is exercised in full), after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us. We expect to use the net proceeds from this offering for working capital and other general corporate purposes, including funding the costs of operating as a public company. See “Use of Proceeds” for additional information.
|
|
Risk factors:
|
You should carefully read “Risk Factors” in this prospectus for a discussion of factors that you should consider before deciding to invest in our securities.
|
|
NASDAQ Capital Market trading symbol for common stock:
|
“CLRX”
|
The number of shares of our common stock to be outstanding after this offering is based on 2,929,954 shares of our common stock outstanding as of September 30, 2014, including shares of common stock subject to repurchase by us, and excludes:
|
·
|
492,147 shares of common stock issuable upon the exercise of outstanding stock options at a weighted-average exercise price of $6.24 per share; |
|
·
|
179,300 shares of common stock issuable upon the vesting of restricted stock units with a weighted average fair value at grant date of $2.37; |
|
·
|
92,888 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $3.15 per share; |
|
·
|
63,671 restricted stock unit awards whose distribution has been deferred; |
|
·
|
27,405 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $2.50 per share; |
|
·
|
251,158 shares available for future issuance under our 2007 Stock Incentive Plan, or the “2007 Plan”; |
|
·
|
shares issuable upon the exercise of warrants sold in this offering;and |
|
·
|
3,705 shares of common stock reserved for future issuance under our Employee Stock Purchase Plan, which expired on July 22, 2014. |
Unless otherwise indicated, all information in this prospectus reflects or assumes the following:
|
·
|
no exercise of outstanding options to purchase common stock or warrants to purchase common stock since September 30, 2014; |
|
·
|
229,300 shares of restricted stock units have vested since September 30, 2014, which includes an additional award of 50,000 shares during the fourth quarter; |
| · | 198,679 shares of common stock were granted to employees and board members during the third quarter; |
|
·
|
92,888 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $3.15 per share expired unexercised on January 14, 2015;
|
|
·
|
37,882 shares are available for future issuance under our 2007 Stock Incentive Plan, or the “2007 Plan”; and |
|
·
|
no exercise by the underwriter of its option to purchase additional shares of common stock and warrants in this offering. |
SUMMARY FINANCIAL DATA
The following summary financial data for the years ended March 31, 2014 and 2013 are derived from our audited financial statements included elsewhere in this prospectus. The summary financial data for the six months ended September 30, 2014 and 2013 have been derived from our unaudited financial statements included elsewhere in this prospectus. These unaudited financial statements have been prepared on a basis consistent with our audited financial statements and, in our opinion, contain all adjustments, consisting only of normal and recurring adjustments, necessary for a fair presentation of such financial data. You should read this data together with our audited financial statements and related notes included elsewhere in this prospectus and the information under the caption “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” Our historical results are not necessarily indicative of our future results, and our operating results for the six-month period ended September 30, 2014 are not necessarily indicative of the results that may be expected for the fiscal year ending March 31, 2015 or any other interim periods or any future year or period.
Income Statement Data:
(in thousands, except per share data)
|
Six Months Ended
|
||||||||||||||||
|
Year Ended March 31,
|
September 30,
|
|||||||||||||||
|
2014*
|
2013*
|
2014**
|
2013**
|
|||||||||||||
|
Revenue
|
$
|
658
|
$
|
300
|
$
|
240
|
$
|
521
|
||||||||
|
Revenue - related party
|
--
|
100
|
-
|
-
|
||||||||||||
|
Total revenue
|
658
|
400
|
240
|
521
|
||||||||||||
|
Cost of revenue
|
158
|
56
|
36
|
36
|
||||||||||||
|
Gross profit
|
500
|
344
|
204
|
485
|
||||||||||||
|
Operating expenses:
|
||||||||||||||||
|
Engineering
|
1,714
|
667
|
1,081
|
748
|
||||||||||||
|
Research and development
|
284
|
536
|
82
|
205
|
||||||||||||
|
Sales and marketing
|
271
|
257
|
153
|
140
|
||||||||||||
|
General and administrative
|
1,819
|
2,979
|
1,197
|
974
|
||||||||||||
|
Total operating expenses
|
4,088
|
4,439
|
2,513
|
2,067
|
||||||||||||
|
Operating loss
|
(3,588
|
)
|
(4,095
|
)
|
(2,309
|
)
|
(1,582
|
)
|
||||||||
|
Other income, net
|
40
|
39
|
9
|
26
|
||||||||||||
|
Loss before income tax benefit
|
(3,548
|
)
|
(4,056
|
)
|
(2,300
|
)
|
(1,556
|
)
|
||||||||
|
Income tax benefit
|
(79
|
)
|
(83
|
)
|
(36
|
)
|
(41
|
)
|
||||||||
|
Loss from continuing operations
|
(3,469
|
)
|
(3,973
|
)
|
(2,264
|
)
|
(1,515
|
)
|
||||||||
|
Gain on sale of discontinued operations, net of taxes
|
267
|
--
|
--
|
267
|
||||||||||||
|
(Loss) income from discontinued operations, net of taxes
|
(112
|
)
|
45
|
--
|
(112
|
)
|
||||||||||
|
Net income from discontinued operations, net of taxes
|
155
|
45
|
--
|
155
|
||||||||||||
|
Net loss
|
$
|
(3,314
|
)
|
$
|
(3,928
|
)
|
$
|
(2,264
|
)
|
$
|
(1,360
|
)
|
||||
|
Net loss per share from continuing operations:
|
||||||||||||||||
|
Basic and diluted
|
$
|
(1.77
|
)
|
$
|
(2.14
|
)
|
$
|
(1.01
|
)
|
$
|
(0.78
|
)
|
||||
|
Net income (loss) per share from discontinued operations:
|
||||||||||||||||
|
Basic and diluted
|
$
|
0.08
|
$
|
0.02
|
$
|
0.00
|
$
|
0.08
|
||||||||
|
Net loss per share:
|
||||||||||||||||
|
Basic and diluted
|
$
|
(1.69
|
)
|
$
|
(2.12
|
)
|
$
|
(1.01
|
)
|
$
|
(0.70
|
)
|
||||
|
Weighted-average shares used in per share computation:
|
||||||||||||||||
|
Basic and diluted
|
1,965
|
1,856
|
2,245
|
1,953
|
||||||||||||
Balance Sheet Data:
(in thousands, except per share data)
|
As of September 30, 2014
|
||||||||
|
Actual
|
As Adjusted
|
|||||||
|
Cash and cash equivalents
|
$
|
1,134
|
$
|
3,818
|
||||
|
Working Capital
|
1,158
|
3,842
|
||||||
|
Promissory note
|
309
|
309
|
||||||
|
Stockholders’ (deficit) equity:
|
||||||||
|
Preferred stock, $0.01 par value; 5,000,000 shares authorized; none issued and outstanding
|
-
|
-
|
||||||
|
Common stock, $0.01 par value; 50,000,000 shares authorized; 2,929,954 shares issued and outstanding actual, 4,903,638 number of shares issued and outstanding pro forma
|
29
|
49
|
||||||
|
Additional paid-in capital
|
132,608
|
135,272
|
||||||
|
Accumulated other comprehensive loss
|
-
|
-
|
||||||
|
Accumulated deficit
|
(130,353
|
)
|
(130,353
|
)
|
||||
|
Total stockholders’ (deficit) equity
|
2,284
|
4,968
|
||||||
|
Total Capitalization
|
$
|
2,593
|
$
|
5,277
|
||||
(1) The as adjusted column in the balance sheet data table above gives effect to the sale and issuance by us of shares of common stock in this offering based upon an assumed public offering price of $1.52 per share, which is the last reported sale price of our common stock on the NASDAQ Capital Market on February 5, 2015, after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us.
You should carefully consider the risks described below, together with all of the other information in this prospectus, before making an investment decision. The risks described below are not the only ones we face. Additional risks we are not presently aware of or that we currently believe are immaterial may also impair our business operations. Our business could be harmed by any of these risks. The trading price of our common stock could decline due to any of these risks, and you may lose all or part of your investment.
Risks Related to Our Business
Our future growth is dependent on the successful development and introduction of new products and enhancements. There can be no assurance that we will be successful in developing and marketing, on a timely basis, new products or product enhancements, or that our new products will adequately address the changing needs of the health care marketplace.
A significant portion of our operating expense is relatively fixed and planned expenditures are based in part on expectations regarding future revenues. As a result, we are generally unable to mitigate the negative impact on margins in the short term. Accordingly, unexpected revenue shortfalls may significantly decrease our operating results from quarter to quarter. As a result, in future quarters our operating results could fall below the expectations of securities analysts or investors, in which event our stock price would likely decrease.
We have a history of losses, expect to incur substantial further losses and may not achieve or maintain profitability in the future, which in turn could further materially decrease the price of our common stock (in thousands).
We had net losses of ($2,264), ($3,314) and ($3,928) for the six months ended September 30, 2014 and the fiscal years ended March 31, 2014 and 2013, respectively. We used cash flows in operating activities of ($1,649), ($2,593) and ($3,838) in these respective periods. We expect to continue to sustain losses for the foreseeable future, which may decrease the price of our common stock. As of September 30, 2014, we had cash and cash equivalents of $1,134. We believed that our existing cash balances were adequate to fund operations only through December 31, 2014.
Failure to raise any needed funds would materially adversely affect us. In consideration of these circumstances, the Company may be forced to consider a merger with or into another company or the liquidation or dissolution of the company, including through a bankruptcy proceeding.
We may not complete our proposed transaction with Medytox Solutions.
On December 6, 2014, we entered into a non-binding letter of intent with Medytox Solutions, Inc. for a potential business combination between the companies. The business combination is subject to, among other things, due diligence, the execution of a definitive agreement, necessary board of director and stockholder approvals and other customary conditions. Our negotiations with Medytox Solutions are at an early stage, and we do not know whether we will enter into a definitive agreement and, if such definitive agreement is entered into, whether the contemplated business combination will be consummated.
If we complete our proposed transaction with Medytox Solutions, your ownership will be significantly diluted and the senior management of Medytox Solutions will control the combined company.
The last reported sale price of our common stock on The NASDAQ Capital Market on February 5, 2015 was $1.52 per share, implying a market capitalization for our company of approximately $4.9 million based on 2,929,954 shares of our common stock outstanding as of September 30, 2014. The last reported sale price of MedytoxSolutions’ common stock on the Over-the-Counter Bulletin Board on February 5, 2015 was $5.00 per share, implying a market capitalization for Medytox Solutions of approximately $145 million based on 29,039,836 shares of Medytox Solutions common stock outstanding as of November 17, 2014.As a result of the significant difference in the relative market capitalizations of our company and Medytox Solutions, we expect that your ownership in the combined company will be significantly diluted. Medytox Solutions is a closely-held corporation, and we expect that members of Medytox Solutions’ senior management will control the combined company in the event that the proposed transaction is completed. In addition, members of Medytox Solutions’ senior management hold preferred stock with rights, preferences and privileges that will rank senior to the common stock of the combined company.
We may incur indebtedness under our Loan and Security Agreement with Medytox Solutions. In the event of a default under the Loan and Security Agreement, we may issue shares of common stock to Medytox Solutions, which will dilute your ownership.
Pursuant to the Letter of Intent, Medytox Solutions agreed to advance certain funding to our company in contemplation of the business combination. On January 16, 2015, we entered into a Loan and Security Agreement with MedytoxSolutions, pursuant to which it is contemplated that Medytox Solutions will loan up to $2,395,644 to our company. We intend to use the proceeds from the Loan and Security Agreement for working capital and general corporate purposes. Amounts borrowed by our company under the Loan and Security Agreement will accrue simple interest at the rate of fifteen percent (15.0%) per year. As of February 4, 2015, we had borrowed $440,715 under the Loan and Security Agreement. The making of additional advances to our company under the Loan and Security Agreement is completely discretionary on the part of MedytoxSolutions. All amounts borrowed under the Loan and Security Agreement mature on December 31, 2015.
Upon the occurrence of an event of default under the Loan and Security Agreement, all or a portion of the then outstanding principal and accrued interest under the Loan and Security Agreement is convertible, in the discretion of MedytoxSolutions, into shares of our common stock at a conversion price equal to the lower of (i) $0.85 or (ii) the average of the bid and ask prices of ourcommon stock on the trading day immediately prior to the date of conversion; provided, however, that the maximum number of shares issuable to Medytox Solutions is 14.9% of the number of shares of common stock then outstanding. The issuance of common stock to Medytox Solutionswould have a dilutive effect on your ownership interest in our company.
We may be required to pay a $1,000,000 termination fee to Medytox Solutions.
On January 16, 2015, we entered into an Agreement with Medytox Solutions, pursuant to which we agreed that in the event we enter into a merger or other sale transaction involving at least thirty-five percent (35.0%) of our shares or assets with a party other than Medytox Solutions, we will pay MedytoxSolutions a $1,000,000 fee. Notwithstanding the foregoing, no fee will be payable to Medytox Solutions in the event (i) Medytox Solutionshas not provided funding to our company of at least $500,000 pursuant to the Loan and Security Agreement or (ii) Medytox Solutionshas not funded an advance requested by us under the Loan and Security Agreement, subject to certain exceptions. The fee may discourage another company from pursuing a strategic transaction with our company.
Our quarterly operating results may continue to fluctuate.
Our revenue and operating results have fluctuated and are likely to continue to fluctuate significantly from quarter to quarter, and we cannot assure you that we will achieve profitability in the future.
Factors that could affect our quarterly operating results include:
|
·
|
fluctuations in demand for our products, and the timing of agreements with strategic partners in the health care marketplace; |
|
·
|
the timing of new products and product enhancements; |
|
·
|
changes in the growth rate of the health care marketplace; |
|
·
|
our ability to control costs, including operations expenses; |
|
·
|
our ability to develop, induce and gain market acceptance for new products and product enhancements; |
|
·
|
changes in the competitive environment, including the entry of new competitors and related discounting of products; |
|
·
|
adverse changes in the level of economic activity in the United States or other major economies in which we do business; |
|
·
|
renewal rates and our ability to up-sell additional products; |
|
·
|
the timing of customer acquisitions; |
|
·
|
the timing of revenue recognition for our sales; and |
|
·
|
future accounting pronouncements or changes in our accounting policies. |
Our future success depends on our ability to retain our key personnel and to successfully integrate them into our management team.
We are dependent on the services of our executive officers, our technical experts and other members of our senior management team, particularly Thomas Mika, our President, Chief Executive Officer, Acting Chief Financial Officer, Secretary and Treasurer. The loss of one or more of these key officers or any other member of our senior management team could have a material adverse effect on us. We may not be able to retain or replace these key personnel, and we may not have adequate succession plans in place. Mr. Mika is subject to employment conditions or arrangements that contain post-employment non-competition provisions. However, these arrangements permit Mr. Mika to terminate his employment with us upon little or no notice and the enforceability of the non-competition provisions is uncertain.
If we are unable to hire, retain and motivate qualified personnel, our business would suffer.
Our future success depends, in part, on our ability to continue to attract and retain highly skilled personnel. The loss of the services of any of our key personnel, the inability to attract or retain qualified personnel or delays in hiring required personnel, particularly in software and biotechnology, may seriously harm our business, financial condition and operating results. Our ability to continue to attract and retain highly skilled personnel will be critical to our future success. Competition for highly skilled personnel is frequently intense, especially in the San Francisco Bay Area. We intend to issue stock options as a key component of our overall compensation and employee attraction and retention efforts. In addition, we are required under U.S. generally accepted accounting principles, or GAAP, to recognize compensation expense in our operating results for employee stock-based compensation under our equity grant programs, which may negatively impact our operating results and may increase the pressure to limit share-based compensation. We may not be successful in attracting, assimilating or retaining qualified personnel to fulfill our current or future needs. Also, to the extent we hire personnel from competitors, we may be subject to allegations that they have been improperly solicited or divulged proprietary or other confidential information.
The personalized healthcare market is rapidly evolving and difficult to predict. If the personalized healthcare market does not evolve as we anticipate or if healthcare market participants do not perceive value in our products, our sales will not grow as quickly as anticipated and our stock price could decline.
We are in a new, rapidly evolving category within the healthcare industry that focuses on using information technology to inform personalized cancer treatment planning. As such, it is difficult to predict important market trends, including how large the personalized healthcare market will be or when and what products customers will adopt. If the market does not evolve in the way we anticipate, if organizations do not recognize the benefit that our products offer, or if we are unable to sell our products to customers, then our revenue may not grow as expected or may decline, and our stock price could decline.
New or existing technologies that are perceived to address personalized cancer treatment planning in different ways could gain wide adoption and supplant our products, thereby weakening our sales and our financial results.
The introduction of products and services embodying new technologies could render our existing products obsolete or less attractive to customers. Other technologies exist or could be developed in the future, and our business could be materially negatively affected if such technologies are widely adopted. We may not be able to successfully anticipate or adapt to changing technology or customer requirements on a timely basis, or at all. If we fail to keep up with technological changes or to convince our customers and potential customers of the value of our products even in light of new technologies, our business, operating results and financial condition could be materially and adversely affected.
We are dependent on a family of products that informs genomic-based medicine.
Our current product offering is limited to a family of products that informs genomic-based medicine using a unique approach of experts and expert systems. We expect to derive a substantial portion of our revenue from this approach and the family of products based on this approach for the foreseeable future. A decline in the price of these products, whether due to competition or otherwise, or our inability to increase sales of these products, would harm our business and operating results. We expect that this concentration of revenue from a single product family comprised of a limited number of products will continue for the foreseeable future. As a result, our future growth and financial performance will depend heavily on our ability to develop and sell enhanced versions of our products. If we fail to deliver product enhancements, new releases or new products that customers want, it will be more difficult for us to succeed.
If we are unable to introduce new products successfully and to make enhancements to existing products, our growth rates would likely decline and our business, operating results and competitive position could suffer.
Our continued success depends on our ability to identify and develop new products and to enhance and improve our existing products, and the acceptance of those products by our existing and target customers. Our growth would likely be adversely affected if:
|
·
|
we fail to introduce these new products or enhancements; |
|
·
|
we fail to successfully manage the transition to new products from the products they are replacing; |
|
·
|
we do not invest our development efforts in appropriate products or enhancements for markets in which we now compete and expect to compete; |
|
·
|
we fail to predict the demand for new products following their introduction to market; or |
|
·
|
these new products or enhancements do not attain market acceptance. |
Any or all of the above problems could materially harm our business and operating results.
Our cash flow is highly variable and may not be sufficient to meet all of our objectives and there is uncertainty about our ability to continue as a going concern.
Our cash and cash equivalents were $1.1 million at September 30, 2014, compared to $2.8 million at September 30, 2013. We have used cash from operations of $2.6 million and $3.8 million for the years ended March 31, 2014 and 2013, respectively. Our existing cash and cash equivalents and expected cash flow from operations will not provide sufficient liquidity to fund our operations and capital expenditures beyond December 31, 2014. Our independent registered public accounting firm concluded that there was substantial doubt about our ability to continue as a going concern as of March 31, 2014. Accordingly, it included an explanatory paragraph to that effect in its report on our March 31, 2014 financial statements.
Until the Company can generate sufficient levels of cash from its operations, we will need to sell equity or debt securities to raise additional funds to continue to operate as a going concern. On January 16, 2015, we entered into a Loan and Security Agreement with MedytoxSolutions, pursuant to which it is contemplated that MedytoxSolutions will loan up to $2,395,644 to our company. As of February 4, 2015, we had borrowed $440,715 under the Loan and Security Agreement. The making of additional advances to our company under the Loan and Security Agreement is completely discretionary on the part of MedytoxSolutions. The perception that we may not be able to continue as a going concern may cause others to choose not to deal with us due to concerns about our ability to meet our contractual obligations and may adversely affect our ability to raise additional capital.
The Company expects to continue to finance future cash needs primarily through proceeds from equity financings and collaborative agreements with strategic partners or through a business combination with a company that has such financing in order to be able to sustain its operations until the Company can achieve profitability and positive cash flows.
Our ability to meet our liquidity needs depends on our ability to achieve revenue targets of between $4 and $5 million annually as well as to reduce expenses. We may have insufficient cash to satisfy our liquidity needs, which could force us to obtain additional debt or equity financing from other sources, to further reduce expenses, or to sell assets. Reducing our expenses could adversely affect our operations. We cannot assure you that we will be able to secure additional debt or equity financing or sell assets on acceptable terms, if at all, and failure to do so could cause us to cease operations. In addition, raising additional equity financing could result in substantial dilution of our equity holders and in the net tangible book value per share of such holdings.
If we are unable to increase market awareness of our company and our products, our revenue may not continue to grow, or may decline.
The personalized healthcare market is nascent, and we have not yet established broad market awareness of our products and services. Market awareness of our value proposition and products will be essential to our continued growth and our success, particularly for the advanced stage treatment segment of the personalized cancer treatment market. If our marketing efforts are unsuccessful in creating market awareness of our company and our products, then our business, financial condition and operating results will be adversely affected, and we will not be able to achieve sustained growth.
We compete in highly competitive markets, and competitive pressures from existing and new companies may adversely impact our business and operating results.
The markets in which we compete are highly competitive. We expect competition to intensify in the future as existing competitors bundle new and more competitive offerings with their existing products and services, and as new market entrants introduce new products into our markets. This competition could result in increased pricing pressure, reduced profit margins, increased sales and marketing expenses and our failure to increase, or the loss of, market share, any of which would likely seriously harm our business, operating results and financial condition. If we do not keep pace with product and technology advances and otherwise keep our product offerings competitive, there could be a material and adverse effect on our competitive position, revenue and prospects for growth.
We compete either directly or indirectly with other medical database solution companies. The principal competitive factors in our markets include, key strategic customer relationships, expert technical personnel, marketplace acceptance of our product, among others.
Many of our current and potential competitors are substantially larger and have greater financial, technical, research and development, sales and marketing, manufacturing, distribution and other resources and greater name recognition. We could also face competition from new market entrants, including our joint-development partners or other current technology partners. In addition, many of our existing and potential competitors enjoy substantial competitive advantages, such as:
|
·
|
longer operating histories; |
|
·
|
the capacity to leverage their sales efforts and marketing expenditures across a broader portfolio of products; |
|
·
|
broader distribution and established relationships with partners; |
|
·
|
access to larger customer bases; |
|
·
|
greater customer support; |
|
·
|
greater resources to make acquisitions; |
|
·
|
larger intellectual property portfolios; and |
|
·
|
the ability to bundle competitive offerings with other products and services. |
As a result, increased competition could result in loss of existing or new customers, price reductions, reduced operating margins and loss of market share. Our competitors also may be able to provide customers with capabilities or benefits different from or greater than those we can provide in areas such as technical qualifications or geographic presence, or to provide end-user customers a broader range of products, services and prices. In addition, some of our larger potential competitors have substantially broader product offerings and could leverage their relationships based on other products or incorporate functionality into existing products to gain business in a manner that discourages users from purchasing our products, including through selling at zero or negative margins, product bundling or closed technology platforms. These larger potential competitors may also have more extensive relationships within existing and potential customers that provide them with an advantage in competing for business with those customers. Our ability to compete will depend upon our ability to provide a better product than our competitors at a competitive price. We may be required to make substantial investments in research, development, marketing and sales in order to respond to competition, and there is no assurance that these investments will achieve any returns for us or that we will be able to compete successfully in the future.
Our limited operating history in the health care market makes it difficult for you to evaluate our current business and future prospects, and may increase the risk of your investment.
Our Company was formed in December 1989 to acquire the operations of the former Tegal Corporation, a division of Motorola, Inc. Until recently, our Company designed, manufactured, marketed and serviced specialized plasma etch systems used primarily in the production of micro-electrical mechanical systems devices, such as sensors, accelerometers and power devices. We exited the semiconductor industry through a series of divestitures in 2010 to 2012. In July 2012, we acquired CollabRx and entered the personalized healthcare market. CollabRx was founded in 2008 to use information technology to inform personalized medicine. Our current management has only been working together with our employee base for a short period of time. This limited operating history in the healthcare market makes financial forecasting and evaluation of our business difficult. Furthermore, because we depend in part on the market’s acceptance of our products, it is difficult to evaluate trends that may affect our business. We have encountered and will continue to encounter risks and difficulties frequently experienced by growing companies in rapidly changing industries. If we do not address these risks successfully, our business and operating results would be adversely affected, and our stock will be adversely affected.
We do not sell our products directly to users and rely instead on contractual relationships with key market participants who derive a benefit from offering our products to their users. Our business development cycles can be long and unpredictable, and our business development efforts require considerable time and expense.
We do not sell our products directly to users. Instead, we form strategic relationships with existing market participants who derive a benefit from offering our products to their users. Our business development efforts involve educating our potential business partners about the use and benefits of our products. Such potential business partners are typically large, well-established companies with long evaluation cycles. We spend substantial time and resources on our business development efforts without any assurance that our efforts will produce any sales. In addition, partnerships are frequently subject to budget constraints, multiple approvals and unplanned administrative, processing and other delays. These factors, among others, could result in long and unpredictable sales cycles. The length of our products’ business development cycles typically range from six to twelve months based on our limited experience, but may be longer as we expand our business development efforts to other segments of the healthcare market. As a result of these lengthy business development time-lines and the uncertain benefit that our partners may derive from offering our products, it is difficult for us to predict when our partners may purchase products from us and as a result, our operating results may vary significantly and may be adversely affected.
Our customers are concentrated and therefore the loss of a significant customer may harm our business.
We generate revenues from a small number of customers. The loss of any of these customers would significantly impact our operating results in future periods. For the three months ended September 30, 2014, three customers accounted for 28.4%, 28.4% and 14.2%, respectively, of the Company’s revenue. For the six months ended September 30, 2014, four customers accounted for 20.9%, 20.9%, 17.4% and 10.4%, respectively, of the Company’s revenue. For the twelve months ended March 31, 2014, one customer, Life Technologies, accounted for 76% of the Company’s revenue and five customers accounted for 96% of the Company’s revenue. In fiscal year 2013, three customers accounted for 100% of our revenues. Specifically, Life Technologies and Everyday Health, Inc. accounted for 62.5% and 12.5%, respectively of the Company’s revenues.
We are exposed to risks associated with contract termination or delay
The software products for which we receive revenue are distributed through third-parties under license or contract, with varying terms. Generally, our agreements with third-parties are subject to termination with notice and/or discretionary termination if certain revenue targets are not achieved. In addition, our agreements generally do not contain revenue or performance guarantees, so our achieved revenues may vary from targets because of changes in strategic direction of our customers, contract termination, or from delays in projected launch dates, over which we have no influence or control. The loss or delay of revenues associated with such contracts may harm our business and cause us to suffer further operating losses.
If our security is breached, our business could be disrupted, our operating results could be harmed, and customers could be deterred from using our products and services.
Our business relies on the secure electronic transmission, storage and hosting of information, including published data and proprietary databases. We face the risk of a deliberate or unintentional incident involving unauthorized access to our computer systems that could result in misappropriation or loss of assets or information, data corruption, or other disruption of business operations. Although we have devoted significant resources to protecting and maintaining the confidentiality of our information, including implementing security and privacy programs and controls, training our workforce, and implementing new technology, we have no guarantee that these programs and controls will be adequate to prevent all possible security threats. We believe that any compromise of our electronic systems, including the unauthorized access, use, or disclosure of information or a significant disruption of our computing assets and networks, would adversely affect our reputation and our ability to fulfill contractual obligations, and would require us to devote significant financial and other resources to mitigate such problems, and could increase our future cyber security costs.
Defects or errors in our software could harm our reputation, result in significant cost to us and impair our ability to market our products.
The software applications underlying our hosted products and services are inherently complex and may contain defects or errors, some of which may be material. Errors may result from our own technology or from the interface of our software with legacy systems and data, which we did not develop. The risk of errors is particularly significant when a new product is first introduced or when new versions or enhancements of existing products are released. The likelihood of errors is increased as a result of our commitment to frequent release of new products and enhancements of existing products.
Material defects in our software could result in a reduction in sales and/or delay in market acceptance of our products. In addition, such defects may lead to the loss of existing customers, difficulty in attracting new customers, diversion of development resources or harm to our reputation. Correction of defects or errors could prove to be impossible or impractical. The costs incurred in correcting any defects or errors or in responding to resulting claims or liability may be substantial and could adversely affect our operating results.
If we experience any failure or interruption in the delivery of our services over the Internet, customer satisfaction and our reputation could be harmed and customer contracts may be terminated.
If we experience any failure or interruption in the delivery of our services over the Internet, customer satisfaction and our reputation could be harmed and lead to reduced revenues and increased expenses.
We may expand our business through new acquisitions in the future. Any such acquisitions could disrupt our business, harm our financial condition and dilute current stockholders’ ownership interests in our company.
We may pursue potential acquisitions of, and investments in, businesses, technologies or products complementary to our business and periodically engage in discussions regarding such possible acquisitions. Acquisitions involve numerous risks, including some or all of the following:
|
·
|
difficulties in identifying and acquiring complementary products, technologies or businesses; substantial cash expenditures; |
|
·
|
incurrence of debt and contingent liabilities, some of which we may not identify at the time of acquisition; |
|
·
|
difficulties in assimilating the operations and personnel of the acquired companies; |
|
·
|
diversion of management’s attention away from other business concerns; |
|
·
|
risk associated with entering markets in which we have limited or no direct experience; |
|
·
|
potential loss of key employees, customers and strategic alliances from either our current business or the target company’s business; and |
|
·
|
delays in customer purchases due to uncertainty and the inability to maintain relationships with customers of the acquired businesses. |
If we fail to properly evaluate acquisitions or investments, we may not achieve the anticipated benefits of such acquisitions, we may incur costs in excess of what we anticipate, and management resources and attention may be diverted from other necessary or valuable activities. An acquisition may not result in short-term or long-term benefits to us. The failure to evaluate and execute acquisitions or investments successfully or otherwise adequately address these risks could materially harm our business and financial results. We may incorrectly judge the value or worth of an acquired company or business. In addition, our future success will depend in part on our ability to manage the growth anticipated with these acquisitions.
Furthermore, the development or expansion of our business or any acquired business or companies may require a substantial capital investment by us. We may not have these necessary funds or they might not be available to us on acceptable terms or at all. We may also seek to raise funds for an acquisition by issuing equity securities or convertible debt, as a result of which our existing stockholders may be diluted or the market price of our stock may be adversely affected.
We may be subject to claims that we or our technologies infringe upon the intellectual property or other proprietary rights of a third party. Any such claims may require us to incur significant costs, to enter into royalty or licensing agreements or to develop or license substitute technology.
We cannot assure you that our software solutions and the technologies used in our product offerings do not infringe patents held by others or that they will not in the future. Any future claim of infringement could cause us to incur substantial costs defending against the claim, even if the claim is without merit, and could distract our management from our business. Moreover, any settlement or adverse judgment resulting from the claim could require us to pay substantial amounts or obtain a license to continue to use the technology that is the subject of the claim, or otherwise restrict or prohibit our use of the technology. Any required licenses may not be available to us on acceptable terms, if at all. If we do not obtain any required licenses, we could encounter delays in product introductions if we attempt to design around the technology at issue or attempt to find another provider of suitable alternative technology to permit us to continue offering the applicable software solution. In addition, we generally provide in our customer agreements that we will indemnify our customers against third-party infringement claims relating to our technology provided to the customer, which could obligate us to fund significant amounts. Infringement claims asserted against us or against our customers or other third parties that we are required or otherwise agree to indemnify may have a material adverse effect on our business, results of operations or financial condition.
We may be unable to adequately enforce or defend our ownership and use of our intellectual property and other proprietary rights.
Our success is heavily dependent upon our intellectual property and other proprietary rights. We rely upon a combination of trademark, trade secret, copyright, patent and unfair competition laws, as well as license and access agreements and other contractual provisions, to protect our intellectual property and other proprietary rights. In addition, we attempt to protect our intellectual property and proprietary information by requiring certain of our employees and consultants to enter into confidentiality, non-competition and assignment of inventions agreements. The steps we take to protect these rights may not be adequate to prevent misappropriation of our technology by third parties or may not be adequate under the laws of some foreign countries, which may not protect our intellectual property rights to the same extent as do the laws of the United States.
Our attempts to protect our intellectual property may be challenged by others or invalidated through administrative process or litigation, and agreement terms that address non-competition are difficult to enforce in many jurisdictions and may not be enforceable in any particular case. Moreover, the degree of future protection of our intellectual property and proprietary rights is uncertain for products that are currently in the early stages of development because we cannot predict which of these products will ultimately reach the commercial market or whether the commercial versions of these products will incorporate proprietary technologies. In addition, there remains the possibility that others will “reverse engineer” our products in order to determine their method of operation and introduce competing products or that others will develop competing technology independently.
If we resort to legal proceedings to enforce our intellectual property rights or to determine the validity and scope of the intellectual property or other proprietary rights of others, the proceedings could be burdensome and expensive, even if we were to prevail. The failure to adequately protect our intellectual property and other proprietary rights may have a material adverse effect on our business, results of operations or financial condition.
If we cease to be a “smaller reporting company” in the future, we will be required to obtain an auditor’s attestation on the effectiveness of our internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act of 2002. Complying with this requirement will increase our accounting costs, and any delay or difficulty in satisfying this requirement could adversely affect our future results of operations and our stock price.
As a smaller reporting company, we are exempt from Section 404(b) of the Sarbanes-Oxley Act of 2002, which requires an independent registered public accounting firm to test the internal control over financial reporting of public companies, and to report on the effectiveness of such controls. If our status as a smaller reporting company changes, we may be required to comply with this auditor attestation requirement. We expect that compliance with this requirement would increase our financial compliance costs and make our audit process more time consuming and costly. In addition, we may in the future discover areas of our internal controls that need improvement, particularly with respect to businesses that we may acquire. If so, we cannot be certain that any remedial measures we take will ensure that we have adequate internal controls over our financial processes and reporting in the future. Any failure to implement required new or improved controls, or difficulties encountered in their implementation could harm our operating results or cause us to fail to meet our reporting obligations. If we are unable to conclude that we have effective internal control over financial reporting, or if it becomes necessary for our independent registered public accounting firm to provide us with an unqualified report regarding the effectiveness of our internal control over financial reporting and it is unable to do so, investors could lose confidence in the reliability of our financial statements. Any failure to implement required new or improved controls, or difficulties or significant costs encountered in the implementation or operation of these controls, could harm our operating results and cause us to fail to meet our financial reporting obligations, which could adversely affect our business and reduce our stock price.
The market for cloud-based and cloud-focused solutions is at a relatively early stage of development, and if it does not develop or develops more slowly than we expect, our business could be harmed.
The market for cloud-based solutions is at an early stage relative to physical appliances and on-premise solutions, and these types of deployments may not achieve or sustain high levels of demand and market acceptance. Many factors may affect the market acceptance of cloud-based and cloud-focused solutions, including:
|
·
|
perceived security capabilities and reliability; |
|
·
|
perceived concerns about the ability to scale operations for large enterprise customers; |
|
·
|
concerns with entrusting a third party to store and manage critical data; and |
|
·
|
the level of configurability or customizability of the solutions. |
If organizations do not establish a presence in the cloud or do not perceive the benefits of our cloud-focused solutions, or if our competitors or new market entrants are able to develop cloud-focused solutions that are or are perceived to be more effective than ours, this portion of our business may not grow further or may develop more slowly than we expect, either of which would adversely affect our business and operating results.
Risks Related to Our Industry
Extensive governmental regulation could require significant compliance costs and have a material adverse effect on the demand for our products.
We do not believe that any of our current or planned products are subject to regulation by the United States Food and Drug Administration (the “FDA”) and other regulatory authorities worldwide. Changes in the level of regulation, including an increase in regulatory requirements, could subject us to regulatory approvals and delays in launching our products. Modifying our products to comply with changes in regulations or regulatory guidance could require us to incur substantial costs. Further, changing regulatory requirements may render our products obsolete or make new products or product enhancements more costly or time consuming than we currently anticipate. Failure by us or our customers to comply with applicable regulations could result in increased regulatory scrutiny and enforcement. If our products fail to comply with government regulations or guidelines, we could incur significant liability or be forced to cease offering our applicable products or services.
If any of our products are deemed to be Medical Devices, thencomplying with medical device regulations on a global scale will be time consuming and expensive, and could subject us to unanticipated and significant delays.
Although the FDA has not determined that any of our products are medical devices that are actively regulated under the Federal Food, Drug and Cosmetic Act and amendments thereto (collectively, the “Act”), the FDA may do so in the future. Other countries have regulations in place related to medical devices that may in the future apply to certain of our products. If any of our products are deemed to be actively regulated medical devices by the FDA or similar regulatory agencies in countries where we do business, we could be subject to extensive requirements governing pre- and post-marketing activities, including pre-market notification clearance. Complying with medical device regulations would be time consuming and expensive, and could result in unanticipated and significant delays in the release of new products or product enhancements. Further, it is possible that these regulatory agencies may become more active in regulating software and medical devices that are used in healthcare. If we are unable to obtain the required regulatory approvals for any such products, our business plans for these products could be delayed or canceled.
The laws governing electronic health data transmissions continue to evolve and are often unclear and difficult to apply. If we or our customers do not maintain the security and privacy of patient records, we may become subject to sanctions by various government entities.
Federal, state, local and foreign laws regulate the confidentiality of patient records and the circumstances under which those records may be released. These regulations govern both the disclosure and use of confidential patient medical record information and require the users of such information to implement specified security and privacy measures. United States regulations currently in place governing electronic health data transmissions continue to evolve and are often unclear and difficult to apply. Laws in non-U.S. jurisdictions may have similar or even stricter requirements related to the treatment of patient information.
In the United States, the Health Insurance Portability and Accountability Act (“HIPAA”) regulations require national standards for some types of electronic health information transactions and the data elements used in those transactions, security standards to ensure the integrity and confidentiality of health information and standards to protect the privacy of individually identifiable health information. Covered entities under HIPAA, which include health care organizations such as our customers, are required to comply with the privacy standards, the transaction regulations and the security regulations. Moreover, the recently enacted Health Information Technology for Economic and Clinical Health (“HITECH”) provisions of the American Recovery and Reinvestment Act of 2009, and associated regulatory requirements, extend many of the HIPAA obligations, formerly imposed only upon covered entities, to business associates as well, which has created additional liability risks related to the privacy and security of individually identifiable health information.
Evolving HIPAA and HITECH-related laws or regulations in the U.S. and data privacy and security laws or regulations in non-U.S. jurisdictions could restrict the ability of our clients to obtain, use or disseminate patient information. This could adversely affect demand for our products if they are not re-designed in a timely manner in order to meet the requirements of any new interpretations or regulations that seek to protect the privacy and security of patient data or enable our clients to execute new or modified health care transactions. We may need to expend additional capital, software development and other resources to modify our products to address these evolving data security and privacy issues.
If commercial third-party payors or government payors fail to provide coverage or adequate reimbursement for certain genomic tests offered by our customers, or if there is a decrease in the amount of reimbursement for our customers’ products or future products they develop, our revenue and prospects for profitability may be harmed.
In both domestic and foreign markets, sales by our customers may depend, in large part, upon the availability of reimbursement from third-party payors. These third-party payors include government healthcare programs such as Medicare, managed care providers, private health insurers, and other organizations. Physicians and patients may not order genomic test services or products from our customers unless commercial third-party payors and government payors pay for all, or a substantial portion, of the list price, and certain commercial third-party payors may not agree to reimburse our customers if Centers for Medicaid and Medicare Services (“CMS”) does not issue a positive coverage decision. There is currently no national coverage decision that determines whether and how certain genomic tests are covered by Medicare. In the absence of a national coverage decision, local Medicare contractors that administer the Medicare program in various regions have some discretion in determining coverage and therefore payment for such tests. Accordingly, if our customers do not receive full or partial reimbursement for their tests, our revenue and results of operations could be adversely affected.
In addition, there is significant uncertainty surrounding whether the use of products that incorporate new technology, such as Next Generation Sequencing (“NGS”)-based tests, for which we provide interpretative analysis, will be eligible for coverage by commercial third-party payors and government payors or, if eligible for coverage, what the reimbursement rates will be for those products. The fact that a diagnostic product has been approved for reimbursement in the past, for any particular indication or in any particular jurisdiction, does not guarantee that such a diagnostic product will remain approved for reimbursement or that similar or additional diagnostic products will be approved in the future. Reimbursement of NGS-based cancer products by commercial third party payors and government payors may depend on a number of factors, including a payor’s determination that products enabled by our molecular information platform are:
|
·
|
not experimental or investigational; |
|
·
|
medically necessary; |
|
·
|
appropriate for the specific patient; |
|
·
|
cost-effective; |
|
·
|
supported by peer-reviewed publications; |
|
·
|
included in clinical practice guidelines; and |
|
·
|
supported by clinical utility studies. |
The United States and foreign governments continue to propose and pass legislation designed to reduce the cost of healthcare. For example, in some foreign markets, the government controls the pricing of many healthcare products. We expect that there will continue to be federal and state proposals to implement governmental controls or impose healthcare requirements. In addition, the Medicare program and increasing emphasis on managed care in the United States will continue to put pressure on product pricing. Cost control initiatives could decrease the price that our customers would receive for any products in the future, which may limit our revenue and profitability.
Risks Related to Our Common Stock and Warrants and This Offering
Management will have broad discretion as to the use of the proceeds from this offering, and we may not use the proceeds effectively.
Our management will have broad discretion in the application of the net proceeds from this offering and could spend the proceeds in ways that you do not agree with or that do not improve our results of operations or enhance the value of our common stock (see “Use of Proceeds”). Our failure to apply these funds effectively could have a material adverse effect on our business and cause the price of our common stock to decline. Additionally, until the net proceeds are used, they may be placed in investments that do not produce significant income or that may lose value.
You will experience immediate and substantial dilution in the net tangible book value per share of the common stock and warrants you purchase.
Since the price per share of our common stock being offered is higher than the net tangible book value per share of our common stock, you will suffer substantial dilution in the net tangible book value of the common stock you purchase in this offering. Based on the assumed public offering price of $1.52 per share, the last reported sale price of our common stock on the NASDAQ Capital Market on February 5, 2015, and after deducting the underwriting discount and estimated offering expenses payable by us, if you purchase shares of common stock in this offering, you will suffer immediate and substantial dilution of $0.71 per share in the net tangible book value of the common stock. See the section entitled “Dilution” in this prospectus for a more detailed discussion of the dilution you will incur if you purchase common stock in this offering.
To the extent that outstanding options or warrants or awards are exercised, you will experience further dilution. As of September 30, 2014, there were:
|
·
|
492,147 shares of common stock issuable upon the exercise of outstanding stock options at a weighted-average exercise price of $6.24 per share; |
|
·
|
179,300 shares of common stock issuable upon the vesting of restricted stock units with a weighted average fair value at grant date of $2.37; |
|
·
|
92,888 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $3.15 per share; |
|
·
|
63,671 restricted stock unit awards whose distribution has been deferred; |
|
·
|
27,405 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $2.50 per share; |
|
·
|
251,158 shares available for future issuance under our 2007 Stock Incentive Plan, or the “2007 Plan”; |
|
·
|
shares issuable upon the exercise of warrants sold in this offering;and |
|
·
|
3,705 shares of common stock reserved for future issuance under our Employee Stock Purchase Plan, which expired on July 22, 2014. |
Unless otherwise indicated, all information in this prospectus reflects or assumes the following:
|
·
|
no exercise of outstanding options to purchase common stock or warrants to purchase common stock since September 30, 2014; |
|
·
|
229,300 shares of restricted stock units have vested since September 30, 2014, which includes an additional award of 50,000 shares during the third quarter; |
|
·
|
92,888 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $3.15 per share expired unexercised on January 14, 2015;
|
|
·
|
37,882 shares are available for future issuance under our 2007 Stock Incentive Plan, or the “2007 Plan”; and |
|
·
|
no exercise by the underwriter of its option to purchase additional shares of common stock and warrants in this offering. |
In addition, we may choose to raise additional capital due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the issuance of these securities could result in further dilution to our stockholders.
In this offering we will sell:
|
·
|
up to $3.0 million of shares, or approximately 61% of our outstanding common stock as of September 30, 2014 (or 70% if the underwriter’s overallotment option is exercised in full), based on the assumed public offering price of $1.52 per share, the last reported sale price of our common stock on the NASDAQ Capital Market on February 5, 2015; and |
|
·
|
warrants to purchase additional shares of common stock. |
This sale and any future sales of a substantial number of shares of our common stock in the public market, or the perception that such sales may occur, could adversely affect the price of our common stock. We cannot predict the effect, if any, that market sales of those shares of common stock or the availability of those shares of common stock for sale will have on the market price of our common stock.
Our common stock could be delisted from NASDAQ.
On November 18, 2014, we were notified by Nasdaq that the bid price of the our common stock closed below the minimum $1.00 per share requirement for continued inclusion under Marketplace Rule 4310(c)(4). In accordance with Marketplace Rule 4310(c)(8)(D), we have180 calendar days, or until May 18, 2015, to regain compliance. If at any time before May 18, 2015, the bid price of our common stock closes at $1.00 per share or more for a minimum of 10 consecutive business days, we will regain compliance with the Rule. If we do not regain compliance by May 18, 2015, an additional 180 days will be granted to regain compliance, so long as we meet The Nasdaq SmallCap Market initial listing criteria (except for the bid price requirement).
On November 20, 2014, we were notified by Nasdaq that the shareholders' equity balance reported on our last Quarterly Report filed with the Securities and Exchange Commission on November 14, 2014 fell below the $2,500,000 minimum requirement for continued listing under the Nasdaq Capital Market's Listing Rule 5550(b)(1). On January 20, 2015, we announced that we received a letter from the NASDAQ Listing Qualifications Staff indicating that, unless we timely request a hearing before the NASDAQ Listing Qualifications Panel, our securities would be delisted from The NASDAQ Capital Market due to our non-compliance with NASDAQ Listing Rule 5550(b)(1). We have requested a hearing before the Panel, at which hearing we will present our plan to evidence compliance with Rule 5550(b)(1), which requires us to maintain a minimum of $2.5 million in stockholders' equity. Our common stock will continue to trade on The NASDAQ Capital Market under the symbol "CLRX" pending completion of the hearing process and the expiration of any extension period granted by the Panel.
In the future, our common stock price or our tangible net worth may fall below the NASDAQ listing requirements, or we may not comply with other listing requirements, with the result being that our common stock might be delisted. If our common stock is delisted, we may list our common stock for trading over-the-counter. Delisting from the NASDAQ could adversely affect the liquidity and price of our common stock and it could have a long-term impact on our ability to raise future capital through the sale of our common stock.
The price of our common stock may fluctuate significantly, and you could lose all or part of your investment.
The closing price of our common stock on The NASDAQ Capital Market has ranged from a high of $5.23 to a low of $0.55 from July 12, 2012, the closing date of our acquisition of CollabRx, through February 5, 2015. The trading price of our common stock may be subject to wide fluctuations in response to various factors, some of which are beyond our control, including:
|
·
|
our quarterly or annual earnings/(losses) or those of other companies in our industry; |
|
·
|
announcements by us or our competitors of significant contracts or acquisitions; |
|
·
|
changes in accounting standards, policies, guidance, interpretations or principles; |
|
·
|
general economic and stock market conditions, including disruptions in the world credit and equity markets; |
|
·
|
the failure of securities analysts to cover our common stock, changes in financial estimates by any securities analysts who follow our company or our failure to meet these estimates or the expectations of investors; |
|
·
|
future sales of our common stock by us or our stockholders; and |
|
·
|
the other factors described in these “Risk Factors.” |
In recent years, the stock market in general has experienced extreme price and volume fluctuations. This volatility has had a significant impact on the market price of securities issued by many companies, including companies in our industry. The price of our common stock could fluctuate based upon factors that have little to do with our performance, and these fluctuations could materially reduce our stock price.
In the past, some companies, including companies in our industry, have had volatile market prices for their securities and have had securities class action suits filed against them. The filing of a lawsuit against us, regardless of the outcome, could have a material adverse effect on our business, financial condition and results of operations, as it could result in substantial legal costs and a diversion of our management’s attention and resources.
Our actual operating results may differ significantly from guidance provided by our management.
Although it has not been our practice in the recent past to do so, we may from time to time release guidance in our quarterly earnings releases, quarterly earnings conference call, or otherwise, regarding our future performance that represent our management’s estimates as of the date of release. This guidance, which includes forward-looking statements, is based on projections prepared by our management. These projections are not prepared with a view toward compliance with published guidelines of the American Institute of Certified Public Accountants, and neither our registered public accountants nor any other independent expert or outside party compiles or examines the projections and, accordingly, no such person expresses any opinion or any other form of assurance with respect thereto.
Projections are based upon a number of assumptions and estimates that, while presented with numerical specificity, are inherently subject to significant business, economic and competitive uncertainties and contingencies, many of which are beyond our control and are based upon specific assumptions with respect to future business decisions, some of which will change. We generally state possible outcomes as high and low ranges which are intended to provide a sensitivity analysis as variables are changed but are not intended to represent such actual results could not fall outside of the suggested ranges. The principal reason that we release guidance is to provide a basis for our management to discuss our business outlook with analysts and investors. We do not accept any responsibility for any projections or reports that may be published by analysts.
Guidance is necessarily speculative in nature, and it can be expected that some or all of the assumptions of the guidance furnished by us will not materialize or will vary significantly from actual results. Accordingly, our guidance is only an estimate of what management believes is realizable as of the date of release. Actual results will vary from our guidance and the variations may be material. In light of the foregoing, investors are urged not to rely upon, or otherwise consider, our guidance in making an investment decision in respect of our common stock. Any failure to successfully implement our operating strategy could result in the actual operating results being different from our guidance, and such differences may be adverse and material.
Provisions of Delaware law and our organizational documents may discourage takeovers and business combinations that our stockholders may consider in their best interests, which could negatively affect our stock price.
Provisions of Delaware law and our certificate of incorporation and bylaws may have the effect of delaying or preventing a change in control of our company or deterring tender offers for our common stock that other stockholders may consider in their best interests. In addition, our board of directors has adopted a shareholder rights plan, or “poison pill,” which has the effect of making it more difficult for a person to acquire control of our company in a transaction not approved by our board of directors.
Our certificate of incorporation authorizes us to issue up to 5,000,000 shares of preferred stock in one or more different series with terms to be fixed by our board of directors. Stockholder approval is not necessary to issue preferred stock in this manner. Issuance of these shares of preferred stock could have the effect of making it more difficult and more expensive for a person or group to acquire control of us, and could effectively be used as an anti-takeover device. Currently there are no shares of our preferred stock issued or outstanding.
Our bylaws provide for an advance notice procedure for stockholders to nominate director candidates for election or to bring business before an annual meeting of stockholders, including proposed nominations of persons for election to our board of directors, and require that special meetings of stockholders be called only by our chairman of the board, chief executive officer, president or the board pursuant to a resolution adopted by a majority of the board.
As a Delaware corporation, we are also subject to provisions of Delaware law, including Section 203 of the Delaware General Corporation law, which prevents some stockholders holding more than 15% of our outstanding common stock from engaging in certain business combinations without approval of the holders of substantially all of our outstanding common stock.
Any provision in our certificate of incorporation, bylaws or Delaware law that has the effect of delaying or deterring a change in control could limit the opportunity for our stockholders to receive a premium for their shares of our common stock, and could also affect the price that some investors are willing to pay for our common stock.
The warrants are speculative in nature.
The warrants do not confer any rights of common stock ownership on their holders, such as voting rights or the right to receive dividends, but rather merely represent the right to acquire shares of common stock at a fixed price for a limited period of time. Specifically, commencing on the date of issuance, holders of the warrants may exercise their right to acquire the common stock and pay an exercise price of $ per share, prior to five years from the date of issuance, after which date any unexercised warrants will expire and have no further value. Moreover, following this offering, the market value of the warrants is uncertain and there can be no assurance that the market value of the warrants will equal or exceed their public offering price. There can be no assurance that the market price of the common stock will ever equal or exceed the exercise price of the warrants, and consequently, whether it will ever be profitable for holders of the warrants to exercise the warrants.
As a public company, we incur significant administrative workload and expenses.
As a public company with common stock listed on The NASDAQ Capital Market, we must comply with various laws, regulations and requirements, including certain provisions of the Sarbanes-Oxley Act of 2002, as well as rules implemented by the SEC and The NASDAQ Stock Market. Complying with these statutes, regulations and requirements, including our public company reporting requirements, continues to occupy a significant amount of the time of our board of directors and management and involves significant accounting, legal and other expenses. We have hired, and anticipate that we will continue to hire, additional accounting personnel to handle these responsibilities, which will increase our operating costs. Furthermore, these laws, regulations and requirements could make it more difficult or more costly for us to obtain certain types of insurance, including director and officer liability insurance, and we may be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. The impact of these requirements could also make it more difficult for us to attract and retain qualified persons to serve on our board of directors, our board committees or as executive officers.
New laws and regulations as well as changes to existing laws and regulations affecting public companies, including the provisions of the Sarbanes-Oxley Act of 2002 and rules adopted by the SEC and by The NASDAQ Stock Market, would likely result in increased costs to us as we respond to their requirements. We are investing resources to comply with evolving laws and regulations, and this investment may result in increased general and administrative expenses and a diversion of management’s time and attention from revenue-generating activities to compliance activities.
We do not currently intend to pay dividends on our common stock and, consequently, your ability to achieve a return on your investment will depend on appreciation in the price of our common stock.
We have never declared or paid any cash dividends on our common stock and do not currently intend to do so for the foreseeable future. We currently intend to invest our future earnings, if any, to fund our growth. Therefore, you are not likely to receive any dividends on your investment in our common stock for the foreseeable future and the success of an investment in shares of our common stock will depend upon any future appreciation in its value. Shares of our common stock may depreciate in value or may not appreciate in value.
This prospectus, including the sections entitled “Prospectus Summary,” “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Business,” contains forward-looking statements, within the meaning of the federal securities laws, that are based on our management’s belief and assumptions and on information currently available to our management. Although we believe that the expectations reflected in these forward-looking statements are reasonable, these statements relate to future events or our future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. Forward-looking statements in this prospectus include, but are not limited to, statements about:
|
·
|
our ability to complete the proposed transaction with Medytox Solutions; |
|
·
|
the role of genomic and other molecular information in the treatment of advanced cancer, including physician’s need for such information; |
|
·
|
our ability to maintain a comprehensive, accurate and current repository of information on genomic and molecular medicine and to continue to ensure our lead over similar information provided by our competitors, including the ability of our knowledgebase and information systems to help physicians treat their patients’ cancers, our first mover advantage in providing comprehensive molecular diagnostic information products on a commercial scale or the sustainability of our competitive advantages; |
|
·
|
our ability to generate revenue from sales of information products to physicians in clinical practice, including our ability to increase adoption of our products and develop new relationships with partners; |
|
·
|
our estimates of the adoption rates of NGS-based, multi-gene cancer panels; |
|
·
|
our estimates of the ability of our laboratory customers to obtain reimbursement for their diagnostic tests, including expectations as to their ability or the amount of time it will take them to achieve successful reimbursement from third-party payors, such as commercial insurance companies and health maintenance organizations, and government insurance programs, such as Medicare and Medicaid; |
|
·
|
our estimates of the penetration and use of our Therapy Finder and CancerRx web-based and mobile products by physicians and the ability of our on-line media partner to successfully sell advertising and sponsorships related to these products; |
|
·
|
our ability to scale our volume of tests from multiple customers, including our capacity to process additional tests at high volume, maintain quality and turn-around time; |
|
·
|
the acceptance of our publications co-authored with our independent expert advisors in peer-reviewed journals or of our presentations at scientific and medical conference presentations; |
|
·
|
our plans and ability to develop and commercialize new information products; |
|
·
|
federal, state, and foreign regulatory requirements, including potential FDA regulation of our information platforms; |
|
·
|
our ability to protect and enforce our intellectual property rights, including our trade secret protected proprietary rights in our information products; |
|
·
|
our anticipated cash needs and our estimates regarding our capital requirements and our needs for additional financing; and |
|
·
|
anticipated trends and challenges in our business and the markets in which we operate. |
In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “should,” “expects,” “intends,” “plans,” “anticipates,” “believes,” “estimates,” “predicts,” “potential,” “continue” or the negative of these terms or other comparable terminology. These statements are only predictions. You should not place undue reliance on forward-looking statements because they involve known and unknown risks, uncertainties and other factors, which are, in some cases, beyond our control and which could materially affect results. Factors that may cause actual results to differ materially from current expectations include, among other things, those listed under “Risk factors” and elsewhere in this prospectus. If one or more of these risks or uncertainties occur, or if our underlying assumptions prove to be incorrect, actual events or results may vary significantly from those implied or projected by the forward-looking statements. No forward-looking statement is a guarantee of future performance. You should read this prospectus and the documents that we reference in this prospectus and have filed with the Securities and Exchange Commission as exhibits to the registration statement, of which this prospectus is a part, completely and with the understanding that our actual future results may be materially different from any future results expressed or implied by these forward-looking statements.
The forward-looking statements in this prospectus represent our views as of the date of this prospectus. We anticipate that subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements at some point in the future, we have no current intention of doing so except to the extent required by applicable law. You should therefore not rely on these forward-looking statements as representing our views as of any date subsequent to the date of this prospectus.
This prospectus also contains statistical data, estimates, and forecasts that are based on independent industry publications, such as those published by McKinsey & Co., World Health Organization, JPMorgan, Goldman Sachs or other publicly available information, as well as other information based on our internal sources. Although we believe that the third-party sources referred to in this prospectus are reliable, neither we nor the underwriter has independently verified the information provided by these third parties. While we are not aware of any misstatements regarding any third-party information presented in this prospectus, their estimates, in particular, as they relate to projections, involve numerous assumptions, are subject to risks and uncertainties, and are subject to change based on various factors, including those discussed under the section titled “Risk Factors” and elsewhere in this prospectus.
We estimate that the net proceeds to us from the sale of shares of common stock and warrants to purchase common stock in this offering will be approximately $2.6 million, based upon an assumed public offering price of $1.52 per share, the last reported sale price of our common stock on the Nasdaq Capital Market on February 5, 2015, and after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us. If the underwriter exercises its option to purchase additional shares and warrants in full, we estimate that our net proceeds will be approximately $3.0 million, after deducting underwriting discounts and commissions and estimated offering expenses payable by us.
In addition, if all of the warrants offered pursuant to this prospectus are exercised in full for cash, we will receive additional cash. However, the warrants contain a cashless exercise provision that permit exercise of warrants on a cashless basis at any time where there is no effective registration statement under the Securities Act of 1933, as amended, covering the issuance of the underlying shares.
We currently intend to use the net proceeds from this offering for working capital and to fund other general corporate purposes, including funding the costs of operating as a public company.
Pending these uses, we may invest the net proceeds in short-term, interest-bearing investment grade securities, certificates of deposit or direct or guaranteed obligations of the U.S. government. We have not determined the amount of net proceeds to be used specifically for such purposes. As a result, management will retain broad discretion over the allocation of net proceeds.
Our common stock is currently traded on the NASDAQ Capital Market under the symbol “CLRX.” The following table sets forth for the periods indicated the high and low sales prices per share of our common stock as reported on the NASDAQ Capital Market:
|
High
|
Low
|
|||||||
|
FISCAL YEAR 2013
|
||||||||
|
First Quarter
|
$
|
3.80
|
$
|
3.10
|
||||
|
Second Quarter
|
5.18
|
3.01
|
||||||
|
Third Quarter
|
5.23
|
3.43
|
||||||
|
Fourth Quarter
|
4.00
|
3.07
|
||||||
|
FISCAL YEAR 2014
|
||||||||
|
First Quarter
|
$
|
3.87
|
$
|
3.06
|
||||
|
Second Quarter
|
4.49
|
3.15
|
||||||
|
Third Quarter
|
4.55
|
3.76
|
||||||
|
Fourth Quarter
|
4.02
|
3.06
|
||||||
|
FISCAL YEAR 2015
|
||||||||
|
First Quarter
|
$
|
3.33
|
$
|
1.86
|
||||
|
Second Quarter
|
2.05
|
1.05
|
||||||
|
Third Quarter
|
1.08
|
0.55
|
||||||
|
Fourth Quarter (through February 3, 2015)
|
1.68
|
0.61
|
||||||
On February 5, 2015, the last reported sale price of our common stock on the NASDAQ Capital Market was $1.52 per share. As of September 30, 2014, we had approximately 120 holders of record of our common stock. The actual number of stockholders is greater than this number of record holders and includes stockholders who are beneficial owners but whose shares are held in street name by brokers and other nominees. This number of holders of record also does not include stockholders whose shares may be held in trust by other entities.
We have never declared or paid any dividends on our capital stock. We currently intend to retain all available funds and any future earnings, if any, to fund the development and expansion of our business and we do not anticipate paying any cash dividends in the foreseeable future. Any future determination to pay dividends will be made at the discretion of our board of directors. Investors should not purchase our common stock with the expectation of receiving cash dividends.
The following table sets forth our cash, cash equivalents and capitalization as of September 30, 2014 as follows:
|
·
|
on an actual basis; and |
|
·
|
on a pro forma basis, giving effect to the sale and issuance by us of up to $3.0 million of common stock and warrants to purchase additional shares of common stock in this offering, at an assumed public offering price of $1.52 per share, which is the last reported sale price of our common stock on the NASDAQ Capital Market on February 5, 2015, after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
(in thousands, except share and per share data)
(Unaudited)
|
As of September 30, 2014
|
||||||||
|
Actual
|
As Adjusted
|
|||||||
|
Cash and cash equivalents
|
$
|
1,134
|
$
|
3,816
|
||||
|
Working Capital
|
1,158
|
3,840
|
||||||
|
Promissory note
|
309
|
309
|
||||||
|
Stockholders’ (deficit) equity:
|
||||||||
|
Preferred stock, $0.01 par value; 5,000,000 shares authorized; none issued and outstanding
|
-
|
-
|
||||||
|
Common stock, $0.01 par value; 50,000,000 shares authorized; 2,929,954 shares issued and outstanding actual, 4,916,709 number of shares issued and outstanding pro forma
|
29
|
49
|
||||||
|
Additional paid-in capital
|
132,608
|
135,270
|
||||||
|
Accumulated other comprehensive loss
|
-
|
-
|
||||||
|
Accumulated deficit
|
(130,353
|
)
|
(130,353
|
)
|
||||
|
Total stockholders’ (deficit) equity
|
2,284
|
4,966
|
||||||
|
Total Capitalization
|
$
|
2,593
|
$
|
5,275
|
||||
You should read the preceding table together with “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” “Description of Capital Stock,” “Description of Warrants” and the financial statements and related notes appearing elsewhere in this prospectus.
The information above is illustrative only and our capitalization following the completion of this offering will be adjusted based on the actual public offering price and other terms of this offering determined at pricing. A $1.0 million decrease in the assumed size of the offering would decrease the amount of cash and cash equivalents, additional paid-in capital, total stockholders’ equity (deficit) and total capitalization on a pro forma basis by approximately $920,000.
The number of shares of our common stock to be outstanding following this offering is based on 2,929,954 shares of our common stock outstanding as of September 30, 2014, and excludes:
|
·
|
492,147 shares of common stock issuable upon the exercise of outstanding stock options at a weighted-average exercise price of $6.24 per share; |
|
·
|
179,300 shares of common stock issuable upon the vesting of restricted stock units with a weighted average fair value at grant date of $2.37; |
|
·
|
92,888 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $3.15 per share; |
|
·
|
63,671 restricted stock unit awards whose distribution has been deferred; |
|
·
|
27,405 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $2.50 per share; |
|
·
|
251,158 shares available for future issuance under our 2007 Stock Incentive Plan, or the “2007 Plan”; |
|
·
|
shares issuable upon the exercise of warrants sold in this offering;and |
|
·
|
3,705 shares of common stock reserved for future issuance under our Employee Stock Purchase Plan, which expired on July 22, 2014. |
Unless otherwise indicated, all information in this prospectus reflects or assumes the following:
|
·
|
no exercise of outstanding options to purchase common stock or warrants to purchase common stock since September 30, 2014; |
|
·
|
229,300 shares of restricted stock units have vested since September 30, 2014, which includes an additional award of 50,000 shares during the fourth quarter; |
| · | 198,679 shares of common stock were granted to employees and board members during the third quarter; |
|
·
|
92,888 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $3.15 per share expired unexercised on January 14, 2015;
|
|
·
|
37,882 shares are available for future issuance under our 2007 Stock Incentive Plan, or the “2007 Plan”; and |
|
·
|
no exercise by the underwriter of its option to purchase additional shares of common stock and warrants in this offering. |
If you invest in our securities in this offering, your ownership interest will be diluted to the extent of the difference between the public offering price per share of our common stock and the net tangible book value per share of our common stock immediately after this offering.
The net tangible book value is determined by dividing our total tangible assets less our total liabilities by the number of shares of common stock outstanding. Our historical net tangible book value (deficit) as of September30, 2014 was $0.504 million, or $0.17 per share. After giving effect to the sale of shares of common stock and warrants by us at the assumed public offering price of $1.52per share and warrant, the last reported sales price of our common stock on the NASDAQ Capital Market on February 5, 2015, our pro forma net tangible book value at September 30, 2014 would have been approximately $3,188, or $0.65 per share. This would represent an immediate increase in the net tangible book value of $0.48 per share to existing stockholders and an immediate dilution of $0.71 per share to investors in this offering. The following table illustrates this per share dilution:
|
Assumed public offering price per share, net
|
$
|
1.36
|
||||||
|
Historical net tangible book value per share as of September 30, 2014
|
$
|
0.17
|
||||||
|
Increase in historical net tangible book value per share attributable to investors in this offering
|
$
|
0.48
|
||||||
|
Pro forma net tangible book value per share after giving effect to this offering
|
$
|
0.65
|
||||||
|
Dilution per share to investors in this offering
|
$
|
0.71
|
||||||
The foregoing illustration does not reflect the potential dilution from the exercise of outstanding options or warrants to purchase shares of our common stock. The foregoing illustration also does not reflect the dilution that would result from the exercise of the warrants sold in the offering.
A $0.10 decrease in the assumed public offering price of $1.52 per share, which is the last reported sale price of our common stock on the NASDAQ Capital Market on February 5, 2014, would decrease the net tangible book value, by $0.07 per share and the dilution to new investors would be $0.64 per share, assuming the size of the offering, as set forth on the cover page of this prospectus, remains the same, and after deducting underwriting discounts and commissions and estimated expenses payable by us.
If the underwriter exercises its over-allotment option in full, at the last reported sale price of our common stock, the net tangible book value would be $[___] per share, and the dilution in net tangible book value per share to investors in this offering would be $[___] per share.
The following table summarizes, on a pro forma basis as of September 30, 2014, the differences between the number of shares of common stock purchased from us, the total consideration and the average price per share paid by existing stockholders and by investors participating in this offering, after deducting estimated underwriting discounts and commissions and estimated offering expenses, at an assumed public offering price of $1.52 per share, the last reported sale price of our common stock on the NASDAQ Capital Market on February 5, 2015.
|
Shares Purchased
|
Total Consideration
|
Ave Price/
|
||||||||||||||||||
|
Number
|
Percent
|
Amount
|
Percent
|
share
|
||||||||||||||||
|
Shares existing stockholders
|
2,929,954
|
59.6
|
%
|
$
|
3,164,350
|
51.3
|
%
|
$
|
1.08
|
|||||||||||
|
Share new investors
|
1,973,684
|
40.4
|
%
|
$
|
3,000,000
|
48.7
|
%
|
$
|
1.68
|
|||||||||||
|
Total
|
4,903,638
|
100.0
|
%
|
$
|
6,164,350
|
100.0
|
%
|
$
|
1.31
|
|||||||||||
The above discussion and table is based on 2,929,954 shares of common stock issued and outstanding as of September 30, 2014 and excludes:
|
·
|
492,147 shares of common stock issuable upon the exercise of outstanding stock options at a weighted-average exercise price of $6.24 per share; |
|
·
|
179,300 shares of common stock issuable upon the vesting of restricted stock units with a weighted average fair value at grant date of $2.37; |
|
·
|
92,888 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $3.15 per share; |
|
·
|
63,671 restricted stock unit awards whose distribution has been deferred; |
|
·
|
27,405 shares of common stock issuable upon the exercise of warrants to purchase common stock with an exercise price of $2.50 per share; |
|
·
|
251,158 shares available for future issuance under our 2007 Stock Incentive Plan, or the “2007 Plan”; |
|
·
|
shares issuable upon the exercise of warrants sold in this offering;and |
|
·
|
3,705 shares of common stock reserved for future issuance under our Employee Stock Purchase Plan, which expired on July 22, 2014. |
To the extent that outstanding options and warrants are exercised, you will experience further dilution. In addition, we may choose to raise additional capital due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans. To the extent that additional capital is raised through the sale of equity or convertible debt securities, the issuance of these securities may result in further dilution to our stockholders.
The net tangible book value is determined by dividing our total tangible assets less our total liabilities by the number of shares of common stock outstanding. Our historical net tangible book value (deficit) as of September 30, 2014 was $0.504 million, or $0.17 per share. After giving effect to the sale of shares of common stock and warrants by us at the assumed public offering price of $1.52 per share and warrant, the last reported sales price of our common stock on the NASDAQ Capital Market on February 5, 2015, our pro forma net tangible book value at September 30, 2014 would have been approximately $3,188, or $0.65 per share. This would represent an immediate increase in the net tangible book value of $0.48 per share to existing stockholders and an immediate dilution of $0.71 per share to investors in this offering.
The following discussion and analysis of our financial condition and results of operations should be read together with financial statements, related notes, and other financial information included elsewhere in this prospectus. This discussion contains forward-looking statements that involve risks and uncertainties. Our actual results could differ materially from those described in, or implied by, the forward-looking statements. Factors that could cause or contribute to those differences include, but are not limited to, those identified below and those discussed above in the section entitled “Risk Factors” included elsewhere in this prospectus.
Overview of our Current Business
CollabRx, Inc. is just entering the commercialization phase of our business. We are focused on developing and delivering content-rich knowledge-based products and services that inform healthcare decision-making, with an emphasis on genomics-based “precision” medicine and big data analytics. Our proprietary content is organized in a knowledge base that expresses the relationship between genetic profiles and therapy considerations including molecular diagnostics, medical tests, clinical trials, drugs, biologics, and other information relevant for cancer treatment planning. We have developed a method for capturing how practicing physicians use this information in the clinical setting, by incorporating within the knowledge base the views of a large network of independent key opinion leaders in medicine and medical research.
We currently deliver our proprietary content to users via web-based applications and services in the “cloud, ” serving physicians and their patients in two settings: (i) at the point-of-care in the clinic and (ii) indirectly, as a part of a genetic test report provided to an ordering physician by a diagnostic testing laboratory, (i.e., the “lab”). Portions of our web-based applications are currently available free to physicians and patients through commercial on-line media partners. The content that we offer to laboratories is based on a “Software as a Service” or SaaS business model, in which our content is provided on a one-time, subscription or per test basis.
At the time of the merger, CollabRx was a development stage company just entering the commercialization phase of business. Following the acquisition, CollabRx recorded initial revenues related to licensing and advertising of its Therapy Finder products on the MedPage Today website. Beginning in the fourth quarter of fiscal 2013 and extending into fiscal 2014, CollabRx recorded revenues related to its fee-for-service activities on behalf of Life Technologies, Inc. (now part of Thermo-Fisher). Initial revenues related to its laboratory products and services were recorded throughout fiscal year 2014 and into the current fiscal year 2015. CollabRx expects to build revenue through the remainder of fiscal 2015 primarily in connection with its Genetic Variant Annotation Service offering to clinical diagnostic laboratories. Significant revenues from advertising and/or sponsorships in connection with its CancerRx mobile app are not expected until next fiscal year beginning April 1, 2015.
During the period following the acquisition on July 12, 2012 and during the balance of fiscal year 2013, which ended March 31, 2013, CollabRx:
|
·
|
Completed the transition of the Company from the former Tegal Corporation to CollabRx, Inc., a data analytics company that uses cloud-based expert systems to inform healthcare decision-making. The Company consolidated operations from Petaluma, CA, and Palo Alto, CA, and completed its relocation into new headquarters in San Francisco, CA; |
|
·
|
Introduced a second-generation Lung Cancer Therapy Finder App, which was made available to 96% of all U.S. oncologists via MedPage Today, a property of Everyday Health, Inc. CollabRx received license fees and a portion of sponsorship revenue associated with the “Oncology Next” webpage on which the Lung Cancer Therapy Finder App was located; |
|
·
|
Initiated activities related to the development of content resources to be used in conjunction with Life Technologies’ global cancer diagnostics development and its laboratory developed test services business under the terms of a multi-year partnership agreement with Life Technologies Inc. (now a part of Thermo-Fisher Scientific, Inc.); and |
|
·
|
Began the development of its laboratory product, later named the “Genetic Variant Annotation Service, or GVA”. |
During fiscal year 2014, which ended March 31, 2014, CollabRx:
|
·
|
Piloted a pre-release version of its GVA Service with two specialty reference labs. Subsequent to the launch of the GVA in August 2013, the Company signed multi-year agreements with Cynvenio Biosystems, Inc. and Quest Diagnostics, Inc.; |
|
·
|
Formed a Pan Cancer (biomarker-focused) molecular oncology editorial board led by Razelle Kurzrock, M.D., serving as its Chief Editor. Dr. Kurzrock is the Senior Deputy Director for Clinical Science at the Moores Cancer Center at UC San Diego. Dr. Kurzrock leads a distinguished group of physicians from leading institutions on the Pan Cancer editorial board, including from the University of Utah, the University of Texas MD Anderson Cancer Center and the University of Maryland Anderson Cancer Center. The Pan Cancer editorial board is differentiated in that it applies a broad molecular oncology perspective in the identification of biomarkers that are clinically actionable in any cancer type; |
|
·
|
Formed a Prostate Cancer board led by E. David Crawford, M.D., serving as its Chief Editor. Dr. Crawford is the distinguished Professor of Surgery, Urology, and Radiation Oncology, and head of the Section of Urologic Oncology at the University of Colorado Anschutz Medical Campus. Dr. Crawford leads a distinguished group of physicians from leading institutions such as Yale University, University of Michigan, Cleveland Clinic, Dana-Farber Cancer Institute, and others; |
|
·
|
Began the development of a Prostate Cancer Therapy Finder, focused initially on neuroendocrine disease; |
|
·
|
Completed the development of a Metastatic Breast Cancer Therapy Finder under the direction of Hope Rugo, M.D., CollabRx’s Breast Cancer Chief Advisor. Dr. Rugo is co-director of the Breast Oncology Clinical Trials Program and is the principal investigator of several clinical trials testing these treatments. She is a professor of medicine at UCSF; and |
|
·
|
Initiated a collaboration with the thoracic oncology program at the University of Chicago Medical Center under the direction of Ravi Salgia, MD, PhD, a professor of medicine and vice chair of translational research at the University of Chicago. |
To date, in fiscal year 2015, CollabRx, has made significant progress in building a strong base for future revenues and establishing a leadership position among oncologists and pathologists in the rapidly emerging area of clinical genetic testing in cancer, including:
|
·
|
Entering into agreements with additional specialty clinical reference laboratories for the GVA Service, including CellNetix Pathology and Laboratories (Seattle, Washington), Genoptix (a Novartis company in Carlsbad, California) and The Jackson Laboratory for Genomic Medicine (Bar Harbor, Maine); |
|
·
|
Completing an agreement with Affymetrix, an industry leader in genomics analysis, to optimize the use of our GVA Service in connection with Affymetrix’s platforms and other industry platforms for analysis of gene copy number variation (CNV) to inform cancer treatment planning. This significant extension of the GVA database opens up new commercial and clinical research customers for the Company; |
|
·
|
Launching CancerRx, an innovative mobile app that combines the Company’s groundbreaking and popular Therapy Finder™ decision support tools in oncology with MedPage Today’s oncology-related news feed. During the week following the launch at the American Society of Clinical Oncology (ASCO) meeting in Chicago at the end of May, more than10,000 cancer healthcare professionals downloaded the app to learn about the latest developments in molecular oncology to help inform the care of their patients; |
|
·
|
Presenting at the ASCO meeting an abstract of a research project done in collaboration with clinical researchers at the University of Chicago Medical Center and University of Wisconsin. The project reinterpreted the findings of several dozen FoundationONE™ reports using the CollabRx GVA to identify new therapeutic options not found in the original reports for a cohort of esophageal cancer patients. This demonstrated the superior database and reporting capability of the GVA when used in planning the treatment of patients with advanced cancer. (FoundationONE™ is a trademark of Foundation Medicine, Inc.); and |
|
·
|
Appointing Paul Billings MD, PhD, FACP, FACMG to its Board of Directors. Dr. Billings is a nationally recognized expert on genomic and precision medicine. He is a board-certified internist and clinical geneticist whose career has been devoted to improving patient care by expanding the use of medically relevant genomic technologies in clinical settings, most recently as Chief Medical Officer of Life Technologies, Inc. (acquired by Thermo Fisher Scientific, Inc. Scientific, Inc. in March 2014). Currently, Dr. Billings serves in multiple roles in industry and government, including as Executive Chairman, Melanoma Diagnostics and a director of Trovagene, DecisionQ, and PAX Neuroscience. Dr. Billings currently serves on the Scientific Advisory Board of the FDA, the Genomic Medicine Advisory Committee at the Department of Veterans Affairs, and the National Academy of Sciences Institute of Medicine’s Roundtable on Genomics. |
On December 6, 2014, we entered into a non-binding letter of intent (the “Letter of Intent”) with Medytox Solutions, Inc. for a potential business combination between the companies. The business combination is subject to, among other things, due diligence, the execution of a definitive agreement, necessary board of director and stockholder approvals and other customary conditions. We cannot assure you that we will enter into a definitive agreement and, if such definitive agreement is entered into, that the contemplated business combination will be consummated.
Pursuant to the Letter of Intent, Medytox Solutions agreed to advance certain funding to CollabRx in contemplation of the business combination. On January 16, 2015, we entered into a Loan and Security Agreement with MedytoxSolutions, pursuant to which it is contemplated that Medytox Solutions will loan up to $2,395,644 to CollabRx. We intend to use the proceeds from the Loan and Security Agreement for working capital and general corporate purposes. Amounts borrowed by our company under the Loan and Security Agreement will accrue simple interest at the rate of fifteen percent (15.0%) per annum. As of February 4, 2015, we had borrowed $440,715 under the Loan and Security Agreement. The making of additional advances to our company under the Loan and Security Agreement is completely discretionary on the part of MedytoxSolutions. All amounts borrowed under the Loan and Security Agreement mature on December 31, 2015. Upon the occurrence of an event of default under the Loan and Security Agreement, all or a portion of the then outstanding principal and accrued interest under the Loan and Security Agreement is convertible, in the discretion of MedytoxSolutions, into shares of our common stock at a conversion price equal to the lower of (i) $0.85 or (ii) the average of the bid and ask prices of ourcommon stock on the trading day immediately prior to the date of conversion; provided, however, that the maximum number of shares issuable to Medytox Solutions is 14.9% of the number of shares of common stock then outstanding. We agreed to secure the payment and performance of our obligations under the Loan and SecurityAgreement by the grant of a security interest in all of our assets.
The Loan Agreement includes representations and warranties of the parties, covenants and agreements regarding the operation of our business while amounts are outstanding under the Loan and Security Agreement, and indemnification provisions in the event of a breach of a representation, warranty, covenant or agreement contained in the Loan and Security Agreement.
Also on January 16, 2015, we entered into an Agreement with MedytoxSolutions, pursuant to which we agreed that in the event we enter into a merger or other sale transaction involving at least thirty-five percent (35.0%) of our shares or assets with a party other than MedytoxSolutions, we will pay MedytoxSolutions a $1,000,000 fee. Notwithstanding the foregoing, no fee will be payable to MedytoxSolutions in the event (i) Medytox Solutions has not provided funding to our company of at least $500,000 pursuant to the Loan and Security Agreement or (ii) MedytoxSolutions has not funded an advance requested by us under the Loan and Security Agreement, subject to certain exceptions.
Company Background
CollabRx (f/k/a Tegal) was formed in December 1989 to acquire the operations of the former Tegal Corporation, a division of Motorola, Inc. Until recently, we designed, manufactured, marketed and serviced specialized systems used primarily in the production of semiconductors and micro-electrical mechanical devices, including integrated circuits, memory devices, sensors, accelerometers and power devices. Beginning in late 2009, we experienced a sharp decline in revenues resulting from the collapse of the semiconductor capital equipment market and the global financial crisis. In a series of transactions from 2010 to 2012, we sold the majority of our operating assets and intellectual property portfolio. During the same time period, our Board of Directors evaluated a number of strategic alternatives, which included the continued operation of the Company as a stand-alone business with a different business plan, a merger with or into another company, a sale of the Company’s remaining assets, and the liquidation or dissolution of the Company. We investigated opportunities within and outside the semiconductor capital equipment industry and evaluated a number of transactions involving other diversified technology-based companies. Throughout this process, we developed and refined our criteria for a business combination, with an eventual focus on the healthcare industry, and specifically information technology and services within the healthcare industry.
On July 12, 2012, we completed the acquisition of a private company called CollabRx, Inc. (the “Merger”), pursuant to an Agreement and Plan of Merger dated as of June 29, 2012 (the “Merger Agreement”). As a result of the Merger, CollabRx became a wholly-owned subsidiary of the Company.
Financial Operations Overview
(In thousands, except per share data)
Comparison of Year Ended March 31, 2014 and 2013
Results of Operations
The following table sets forth certain financial items for the years indicated:
|
Year Ended March 31,
|
Change
|
|||||||||||||||
|
2014
|
2013
|
$
|
%
|
|||||||||||||
|
Revenue
|
$
|
658
|
$
|
300
|
$
|
358
|
119.3
|
%
|
||||||||
|
Revenue - related party
|
--
|
100
|
(100
|
)
|
-100.0
|
%
|
||||||||||
|
Total revenue
|
658
|
400
|
258
|
64.5
|
%
|
|||||||||||
|
Cost of revenue
|
158
|
56
|
102
|
182.1
|
%
|
|||||||||||
|
Gross profit
|
500
|
344
|
156
|
45.3
|
%
|
|||||||||||
|
Operating expenses:
|
||||||||||||||||
|
Engineering
|
1,714
|
667
|
1,046
|
156.8
|
%
|
|||||||||||
|
Research and development
|
284
|
536
|
(252
|
)
|
-47.0
|
%
|
||||||||||
|
Sales and marketing
|
271
|
257
|
14
|
5.4
|
%
|
|||||||||||
|
General and administrative
|
1,819
|
2,979
|
(1,160
|
)
|
-38.9
|
%
|
||||||||||
|
Total operating expenses
|
4,088
|
4,439
|
(351
|
)
|
-7.9
|
%
|
||||||||||
|
Operating loss
|
(3,588
|
)
|
(4,095
|
)
|
507
|
-12.4
|
%
|
|||||||||
|
Other income, net
|
40
|
39
|
1
|
2.6
|
%
|
|||||||||||
|
Loss before income tax benefit
|
(3,548
|
)
|
(4,056
|
)
|
508
|
-12.5
|
%
|
|||||||||
|
Income tax benefit
|
(79
|
)
|
(83
|
)
|
4
|
-4.8
|
%
|
|||||||||
|
Loss from continuing operations
|
(3,469
|
)
|
(3,973
|
)
|
504
|
-12.7
|
%
|
|||||||||
|
Gain on sale of discontinued operations, net of taxes
|
267
|
--
|
267
|
|||||||||||||
|
(Loss) income from discontinued operations, net of taxes
|
(112
|
)
|
45
|
(157
|
)
|
-348.9
|
%
|
|||||||||
|
Net income from discontinued operations, net of taxes
|
155
|
45
|
110
|
244.4
|
%
|
|||||||||||
|
Net loss
|
$
|
(3,314
|
)
|
$
|
(3,928
|
)
|
$
|
614
|
-15.6
|
%
|
||||||
|
Net loss per share from continuing operations:
|
||||||||||||||||
|
Basic and diluted
|
$
|
(1.77
|
)
|
$
|
(2.14
|
)
|
||||||||||
|
Net income per share from discontinued operations:
|
||||||||||||||||
|
Basic and diluted
|
$
|
0.08
|
$
|
0.02
|
||||||||||||
|
Net loss per share:
|
||||||||||||||||
|
Basic and diluted
|
$
|
(1.69
|
)
|
$
|
(2.12
|
)
|
||||||||||
|
Weighted-average shares used in per share computation:
|
||||||||||||||||
|
Basic and diluted
|
1,965
|
1,856
|
||||||||||||||
Revenue
While we have partnership agreements for advertising revenue related to our web-based and mobile applications, we have not yet generated significant revenue from that source.
Immediately prior to the acquisition of CollabRx, our sole source of revenue was from management activities related to Sequel Power. Sequel Power was a related party. As of March 31, 2013, we terminated our management services contract with Sequel Power and canceled outstanding warrants in consideration for the forgiveness of related outstanding accounts receivable balance and our interest in Sequel Power. We are no longer involved in supporting the activities of Sequel Power through our direct management efforts.
Our Therapy Finder™ web-based application serves as an initial user-interface to the underlying knowledge base. It is available free of charge on our website. A professional version is offered to registered physicians through MedPage Today, an offering of Everyday Health, Inc. Our agreement with Everyday Health provides for an annual license fee payable to our company and sharing of sponsorships and advertising revenue generated by Everyday Health.
The Company also provides clinical interpretation of lab results regarding genetic variants present in human tumor biopsies, and these interpretative reports are sold directly to diagnostic labs that perform molecular testing on patients. Our “Genetic Variant Application” or “GVA” is compiled dynamically by our software platform to provide specific insights to a patient’s diagnostic test results on a test-by-test basis. The GVA results are provided to laboratories in a variety of forms, including with a front-end user Interface or UI or directly integrated into a customer’s laboratory information management system or LIMS. We deliver the GVA product via a cloud-based variation on a SaaS business model, and we receive payment directly from the diagnostic laboratory, typically on a per-test basis.
The Company also provides specific customer data reports which are not accessible through usage-based or subscription service arrangements to be a separate deliverable, as in the case with Life Technologies. We do not allow these reports to be re-sold and we do not offer a right of return once the report is delivered.
Revenue for fiscal year 2014 increased by $358 to $658 compared $300 for to fiscal year 2013. The increase relates to our acquisition of CollabRx and the generation of revenue in connection with commercial agreements.
As a percentage of total revenue for both the fiscal years 2014 and 2013, international sales were 0%. We expect our international sales will account for a significant portion of future revenue once our commercialization activities become more widely accepted.
Gross Profit
Gross profit for the year ended March 31, 2014 increased $156 to $500 from our gross profit of $344 for the year ended March 31, 2013. The increase in our gross profit for the year ended March 31, 2014 was generated by the continuing initial commercialization activities of CollabRx represented by agreements with Life Technologies, Inc. and Everyday Health, Inc. and new customers.
Our gross profit margin for the year ended March 31, 2014 was 76% and primarily reflects the amortization of our product specific software, which was included in the CollabRx merger. Our gross margin for the year ended March 31, 2013 was 86%, as 25% of revenues in that period were management services revenues and no costs were incurred to record this revenue.
At the present time our core operations consist of the development and commercial application of the CollabRx technology and content. We offer cloud-based expert systems that provide clinically relevant interpretive knowledge to institutions, physicians, researchers and patients for genomics-based medicine in cancer.
Engineering
Following the acquisition of CollabRx, engineering expenses consist primarily of salaries. Our engineering expenses increased to $1,714 in fiscal 2014 from $667 in fiscal 2013, and resulted from the CollabRx acquisition and the employees retained for those operations. A portion of certain employee related engineering expenses are re-categorized from engineering to research and development. (See “Research and Development” below.) The increase in engineering expenses in fiscal year 2014 compared to fiscal year 2013 reflected compensation paid to scientists and engineers that became our employees in connection with the CollabRx acquisition, effective July 12, 2012, as well as additional hires since the acquisition date. The increase was due primarily to salary and stock compensation expense as the Company had only three quarters of engineering expenses in fiscal year 2013 compared to four quarters of Engineering expenses in fiscal year 2014. In addition, the change reflects increases in recruiting, rent and subscription expenses.
Prior to the CollabRx acquisition, the Company had exited from our core historical Deep Reactive Ionic Etching (“DRIE”) operations.
Research and Development
We define “engineering” as those development activities that are related to products, content or services which have been offered for sale or for which we have been paid. We define research and development (“R&D”) as those development activities which are not related to products which have been offered for sale or for which we have been paid.
The efforts of our engineering group include supporting existing product offerings as well as developing future product offerings. We have segregated these expenses and reported expenses that make up the total R&D expenses for fiscal years 2014 and 2013, respectively.
We include all of those employees who work both on engineering activities and R&D activities in the headcount within engineering and allocate the expense to R&D, as categorized above. The efforts of the engineering group include supporting existing product offerings as well as developing future product offerings. Consequently, such expenses are segregated. The decrease in R&D expenses of $252 in fiscal year 2014 compared to fiscal year 2013 is related to the efforts of engineering being directed to supporting new customer offerings.
As a result of the sale of the Company’s DRIE related assets, and in accordance with generally accepted accounting principles, the DRIE business operation, including related and ongoing minor R&D expenses, have all been reclassified to discontinued operations. For the fiscal years ended March 31, 2014 and 2013, respectively, the Company’s discontinued R&D expenses related to the NLD product line, the assets of which were held for sale and subsequently sold to third parties, were included in discontinued operations.
For the fiscal year ended March 31, 2014, we had no employees dedicated to R&D. A former employee was responsible, on a contract basis, for managing the activities related to the sale of our intellectual property. The Company sold the last of its patents in the fiscal year 2014 and has no other intellectual property related to discontinued operations.
Sales and Marketing
With the acquisition of CollabRx, our sales and marketing expenses increased and consist primarily of salaries. Our sales and marketing expenses increased to $271 in fiscal 2014 from $257 in fiscal 2013. The increase was due primarily to salary and stock compensation expense as the Company had only three quarters of sales and marketing expenses in Sales and Marketing in fiscal year 2013 compared to four quarters in fiscal year 2014. The increase in salary and stock compensation expense was offset by a decrease in outside services.
General and Administrative
General and administrative expenses consist of salaries, legal, accounting and related administrative services and expenses associated with general management, finance, information systems, human resources and investor relations activities. General and administrative expenses decreased to $1,819 in fiscal year 2014 compared to $2,979 for fiscal year 2013. The decrease was due primarily to the acquisition costs and cash bonuses for key employees paid in the prior year. Acquisition costs related to CollabRx included expenses for legal, accounting and consulting services.
Unconsolidated Affiliate
On March 31, 2013, Sequel Power irrevocably assigned and transferred unto the Company for cancelation the balance of its Warrants representing the right to purchase 44,578 shares of the Company’s common stock. In exchange, the Company agreed to waive receivables related to certain fees earned under its Services Agreement with Sequel Partners and its 25% ownership interest in Sequel Power. In addition, effective March 31, 2013, the Company terminated its management services agreement with Sequel Power.
Other Income (Expense), net
Other income (expense), net consists of the change in fair value of the common stock warrant liability, the interest earned on our NanoVibronix investment, and the interest accrued on our promissory note payable.
Discontinued Operations
Discontinued operations consists of interest income from accounts related to discontinued operations, gains and losses on the disposal of fixed assets of discontinued operations, and gains and losses on foreign exchange, as well as the reclassification of net expenses associated with our exit from our historical core operations.
During fiscal 2014, we recognized $365 from the sale of the last of our NLD patents. As these assets were internally developed, there was a corresponding zero book value. The NLD revenue was offset by related expenses of $98, resulting in a net gain, net of taxes, of $267. With this sale, the Company has no other intellectual property related to discontinued operations. Discontinued operations also included expenses related to the final closing of former foreign subsidiaries. In fiscal 2014, the Company recognized a reclassification out of accumulated other comprehensive loss and into Loss from Discontinued Operations, net of taxes. The reclassification is related to the recognition of a non-cash loss of $142 from the of foreign exchange differences from its former Tegal foreign subsidiaries, primarily as a result of the final closing of the former Tegal German subsidiary. The Company received permission to close the German subsidiary in June 2013. No further audits or reviews are anticipated by foreign taxing authorities. The Company also recognized a cash gain of $20 in discontinued operations as a result of final closing of bank accounts in its Italian subsidiary and a federal tax refund regarding discontinued operations, and a net $4 non cash gain related to the write off of discontinued assets and liabilities in its foreign subsidiaries. An $8 tax refund from Sonoma county related to an unsecured property tax refund for 2010/2011 was also recognized in discontinued operations.
In fiscal 2013, discontinued operations included a gain resulting from the net settlement of legal expenses related to closing a foreign subsidiary (for which a higher amount of legal expense had been accrued in the prior fiscal year), offset by R&D expenses included in discontinued operations.
Total revenue from discontinued operations for fiscal years 2014 and 2013 was $0. The total income from discontinued operations, including income tax expense, was $155 and $45, for the same years, respectively.
The Company did not record any severance charges for either fiscal year 2014 or fiscal year 2013. We had no outstanding severance liability as of March 31, 2014.
Income Taxes
As a result of the stock purchase of CollabRx during the fiscal year ended March 31, 2013, we had no tax basis in the intangible assets acquired. During the twelve months ended March 31, 2013, we recognized $83 in tax benefit as a result of this difference. During the year ended March 31, 2014, we recognized $81 in tax benefit as a result of this difference.
During the year ended March 31, 2014, there was no income tax expense or benefit for federal and state income taxes reflected in our consolidated statements of operations due to our net loss and a valuation allowance on the resulting deferred tax asset.
In each fiscal 2014 and 2013, our effective tax rate was (2%).
As of March 31, 2014, the Company had net operating loss carryforwards of approximately $121.5 million and $65.2 million for federal and state tax purposes, respectively. The federal net operating loss carryforward will begin to expire in the year ending March 31, 2020 and the state of California net operating loss carryforward began to expire in the year ended March 31, 2013. As of March 31, 2014, the Company also had research and experimentation credit carryforwards of $1.4 million and $0.9 million for federal and state income tax purposes, respectively. A portion of the federal credit began to expire in the year ended March 31, 2012 and the state of California will never expire under current law. Net operating loss carryforwards and R&D credits can only offset 90% of state taxable income.
As of March 31, 2013, the Company had net operating loss carryforwards of approximately $111.8 million and $64.9 million for federal and state tax purposes, respectively. The federal net operating loss carryforward will begin to expire in the year ended March 31, 2020 and the state of California net operating loss carryforward began to expire in the year ended March 31, 2013. At March 31, 2013, the Company also had research and experimentation credit carryforwards of $1.3 million and $0.8 million for federal and state income tax purposes, respectively. A portion of the federal credit began to expire in the year ended March 31, 2012 and the state of California will never expire under current law. Net operating loss carryforwards and R&D credits can only offset 90% of taxable income.
Comparison of the Three and Six Months Ended September 30, 2014 and 2013
Results of Operations
The following table sets forth certain financial items for the periods indicated:
(in thousands, except per share data)
|
Three Months Ended
September 30,
|
Six Months Ended
September 30,
|
|||||||||||||||
|
2014 **
|
2013 **
|
|
2014 **
|
|
2013 **
|
|||||||||||
|
Revenue
|
$
|
176
|
$
|
251
|
$
|
240
|
$
|
521
|
||||||||
|
Cost of revenue
|
18
|
18
|
36
|
36
|
||||||||||||
|
Gross profit
|
158
|
233
|
204
|
485
|
||||||||||||
|
Operating expenses:
|
||||||||||||||||
|
Engineering
|
539
|
516
|
1,081
|
748
|
||||||||||||
|
Research and development
|
32
|
31
|
82
|
205
|
||||||||||||
|
Sales and marketing
|
73
|
73
|
153
|
140
|
||||||||||||
|
General and administrative
|
553
|
485
|
1,197
|
974
|
||||||||||||
|
Total operating expenses
|
1,197
|
1,105
|
2,513
|
2,067
|
||||||||||||
|
Operating loss
|
(1,039
|
)
|
(872
|
)
|
(2,309
|
)
|
(1,582
|
)
|
||||||||
|
Other income, net
|
2
|
16
|
9
|
26
|
||||||||||||
|
Loss before income tax benefit
|
(1,037
|
)
|
(856
|
)
|
(2,300
|
)
|
(1,556
|
)
|
||||||||
|
Income tax benefit, net
|
(21
|
)
|
(20
|
)
|
(36
|
)
|
(41
|
)
|
||||||||
|
Loss from continuing operations
|
(1,016
|
)
|
(836
|
)
|
(2,264
|
)
|
(1,515
|
)
|
||||||||
|
Gain on sale of discontinued operations, net of taxes
|
--
|
267
|
--
|
267
|
||||||||||||
|
Income/(loss) from discontinued operations, net of taxes
|
--
|
6
|
--
|
(112
|
)
|
|||||||||||
|
Net income from discontinued operations, net of taxes
|
--
|
273
|
--
|
155
|
||||||||||||
|
Net loss
|
$
|
(1,016
|
)
|
$
|
(563
|
)
|
$
|
(2,264
|
)
|
$
|
(1,360
|
)
|
||||
|
Net loss per share from continuing operations:
|
||||||||||||||||
|
Basic and diluted
|
$
|
(0.35
|
)
|
$
|
(0.43
|
)
|
$
|
(1.01
|
)
|
$
|
(0.78
|
)
|
||||
|
Net income per share from discontinued operations:
|
||||||||||||||||
|
Basic and diluted
|
$
|
-
|
$
|
0.14
|
$
|
-
|
$
|
0.08
|
||||||||
|
Net loss per share:
|
||||||||||||||||
|
Basic and diluted
|
$
|
(0.35
|
)
|
$
|
(0.29
|
)
|
$
|
(1.01
|
)
|
$
|
(0.70
|
)
|
||||
|
Weighted-average shares used in per share computation:
|
||||||||||||||||
|
Basic and diluted
|
2,929
|
1,953
|
2,245
|
1,953
|
||||||||||||
** Unaudited
Revenue
Revenue for the three and six month periods ended September 30, 2014 decreased by $75 and $281, respectively, compared to the three and six month periods ended September 30, 2013. Revenues in fiscal 2014 were derived primarily from the Company’s first multiple-element arrangement with a strategic customer. This arrangement began in fiscal year 2013 and expired in fiscal year 2014. Revenues in fiscal 2015 were derived from multiple customer SaaS service agreements, a royalty agreement, advertising revenues and testing fees.
As a percentage of total revenue for the three and six months ended September 30, 2014, international sales were an immaterial portion of total revenues. As a percentage of total revenue for the three and six months ended September 30, 2013, international sales were 0%.
Gross Profit
Gross profit for the three and six months ended September 30, 2014 decreased $75 and $281, respectively, compared to the three and six months ended September 30, 2013. Our gross profit reflects the amortization of the Company’s product specific software, which was included in the CollabRx acquisition. Any engineering expenses related to revenue are also included in cost of revenue. For the three and six months ended September 30, 2014 and 2013, respectively, there were no additional engineering expenses included in cost of revenue.
Our gross margins for the three and six months ended September 30, 2014 were 89.8% and 85.0%, respectively. Our gross margins for the three and six months ended September 30, 2013 was 92.8% and 93.1% respectively. These periods included revenue solely derived from our genomics based information products. The amortization of acquired software is included in cost of goods.
Engineering
Engineering expenses consist primarily of salaries, and those salaries and related expenses are assigned to either Engineering or R&D based on the specific projects that the staff is working on during the quarter. The increase in Engineering expense of $23 and $333 for the three and six months ended September 30, 2014, compared to the same period in 2013, reflected higher employee related expenses due to additional headcount and a greater level of effort focused on existing products than on products that had not yet been offered for sale.
Research and Development
The increase of R&D expense of $1 and the decrease of $123 for the three and six month periods ended September 30, 2014, respectively, compared to the same periods in 2013 reflects the focus of development activities on products offered for sale, as opposed to those that may be offered in the future. The launch of the Genetic Variant Annotation Service in August 2013 significantly lowered the amount of effort being devoted to future products. Extensions or improvements to the Therapy Finders, CancerRx mobile app and the GVA, along with fee-for-service development activities are all assigned as Engineering expenses rather than R&D.
Sales and Marketing
Sales and marketing expenses consist primarily of salaries. For the three months ended September 30, 2014 and 2013, respectively, sales and marketing expenses were flat. For the six months ended September 30, 2014 and 2013, respectively, the increase of $13 resulted primarily from increased expenses related to medical conference attendance.
General and Administrative
The increase in general and administrative expenses of $68 and $233 for the three and six month periods ended September 30, 2014, compared to the same period in 2013 was due primarily to higher expenses related to consultants, investor relations and presentations, as well as higher expenses for director compensation, stock compensation expense and corporate taxes.
Other Income, net
Other income, net primarily consists of the interest earned on our NanoVibronix investment.
Income Taxes
As a result of the acquisition of CollabRx by stock purchase, the Company had no tax basis in the intangible assets acquired. During the three and six months ended September 30, 2014, the Company recognized $21 and $41 in each period, respectively, in tax benefit as a result of this difference. The Company also recognized $20 and $41 in each of three and six month periods ended September 30, 2013 in tax benefit as a result of this difference.
Due to our net loss and the aforementioned valuation allowance on the resulting deferred tax asset, the Company recognized no federal or state income taxes in our condensed consolidated statements of operations for the three and six months ended September 30, 2014 and 2013, respectively.
The Company did however recognize $5 for city taxes and the annual minimum amount due for state income taxes in the six months ended September 30, 2014.
As of March 31, 2014, the Company had net operating loss carryforwards of approximately $121.5 million and $65.2 million for federal and state tax purposes, respectively. The federal net operating loss carryforward will begin to expire in the year ending March 31, 2020 and the state of California net operating loss carryforward began to expire in the year ended March 31, 2013. As of March 31, 2014, the Company also had research and experimentation credit carryforwards of $1.4 million and $0.9 million for federal and state income tax purposes, respectively. A portion of the federal credit began to expire in the year ended March 31, 2012 and the state of California will never expire under current law. Net operating loss carryforwards and R&D credits can only offset 90% of taxable state income.
Discontinued Operations
The Company no longer has any assets or liabilities associated with discontinued operations as of the end of fiscal year 2014.
In the six months ended September 30, 2013, the Company completed the final closing of bank accounts in its Italian subsidiary. It also recognized a net $4 non cash gain related to the write off of discontinued assets and liabilities in its foreign subsidiaries, and recognized a reclassification out of accumulated other comprehensive loss and into Loss from Discontinued Operations, net of taxes. The reclassification is related to the recognition of a non-cash loss of $142 from the of foreign exchange differences from its former Tegal foreign subsidiaries, primarily as a result of the final closing of the former Tegal German subsidiary. The Company received permission to close the German subsidiary in June 2013. No further audits or reviews are anticipated by foreign taxing authorities.
With the closure of the former Tegal’s foreign subsidiaries and the sale of the Company’s last two patent lots in fiscal year 2014, the Company has no other activities or assets related to discontinued operations.
Contractual Obligations
The following summarizes our contractual obligations as of September 30, 2014, and the effect such obligations are expected to have on our liquidity and cash flows in future periods (in thousands).
|
Contractual obligations:
|
Total
|
Less than
1 Year
|
1-3 Years
|
3-5 Years
|
After
5 Years
|
|||||||||||||||
|
Promissory note payable
|
$
|
500
|
$
|
167
|
$
|
333
|
$
|
-
|
$
|
-
|
||||||||||
|
Interest due on convertible promissory note payable
|
71
|
41
|
30
|
-
|
-
|
|||||||||||||||
|
Non-cancelable operating lease obligations
|
371
|
125
|
246
|
-
|
-
|
|||||||||||||||
|
Total contractual cash obligations
|
$
|
942
|
$
|
333
|
$
|
609
|
$
|
-
|
$
|
-
|
||||||||||
Most leases provide for the Company to pay real estate taxes and other maintenance expenses. Rent expense for operating leases related to discontinued operations, net of sublease income, was $0 for the periods presented. Rent expense for operating leases related to continuing operations was $33 and $65 for the three and six month periods ended September 30, 2014, respectively. Rent expense for operating leases related to continuing operations was $32 and $73 for the three and six month periods ended September 30, 2013, respectively. The Company has no sublease income for the periods presented.
Certain of our past sales contracts included provisions under which customers would be indemnified by us in the event of, among other things, a third party claim against the customer for intellectual property rights infringement related to our products. There are no limitations on the maximum potential future payments under these guarantees. We have accrued no amounts in relation to these provisions as no such claims have been made, and we believe we have valid, enforceable rights to the intellectual property embedded in our products.
In addition to the non-cancelable operation lease obligations above, as part of the CollabRx acquisition, we assumed $500 of existing CollabRx indebtedness through the issuance of promissory notes. The principal amount of the promissory notes is payable in equal installments on the third, fourth and fifth anniversaries of the closing date of the Merger, along with the accrued but unpaid interest as of such dates.
Liquidity and Capital Resources
For the six months ended September 30, 2014 and the fiscal year ended March 31, 2014, we financed our operations from existing cash on hand and the net proceeds from the sale of discontinued assets, as disclosed in prior filings, and the net proceeds raised from an underwritten public offering which closed on June 25, 2014. Net cash used in operating activities during the six months ended September 30, 2014 was $1,649. The primary changes in our cash flow statement for the six months ended September 30, 2014 compared to the corresponding period in the prior fiscal year were due to our net loss of $2,264, partially offset by changes in stock-based compensation expense, amortization of intangibles, deferred financing expenses of the Company’s recent round of new financing and changes in accounts receivable. Net cash used in operating activities during the six months ended September 30, 2013 was $1,237, due primarily to our net loss of $1,360 and changes in assets and liabilities of discontinued operations, partially offset by stock compensation expense and amortization of intangibles.
The condensed consolidated financial statements contemplate the realization of assets and the satisfaction of liabilities in the normal course of business for the foreseeable future. We incurred net losses of $2,264 and $1,360 for the six months ended September 30, 2014 and 2013, respectively. We believed that our existing cash was be adequate to fund our operations only through December 31, 2014.
On June 25, 2014, the Company closed an underwritten public offering of 913,500 shares of its common stock, offered at a public offering price of $2.00 per share. Gross proceeds to CollabRx from this offering were $1,827 before deducting the underwriting discount and other estimated offering expenses payable by CollabRx. CollabRx has used the net proceeds from the offering for general corporate purposes, including development of our products and services, general and administrative expenses and working capital. Aegis Capital Corp. acted as the sole book-running manager for the offering. In addition to the offering of 913,500 shares of common stock, 27,405 warrants to purchase shares of common stock were issued in connection with this offering. These warrants have an exercise price of $2.50 per share and are not exercisable until June 24, 2015 and expire June 24, 2020. Such securities could potentially dilute earnings per share in future periods. Previously deferred financing expenses incurred to raise equity capital related to financing transactions prior to the completion of the financing, as well as other related expenses incurred in the current period, were recognized in the current period and offset the gross proceeds raised. Total underwriting discount and financing expenses were $480. The Company netted $1,347 after underwriting expenses.
The Company also received $23 from the sale of 7,101 shares through an at-the-market financing facility through Cantor Fitzgerald.
Without additional capital, our recurring losses from operations raise substantial doubt about our ability to continue as a going concern. Even though the Company has entered into the aforementioned agreement with Medytox, it must still prove its ability to generate sufficient levels of cash from its operations. The Company expects to continue to finance future cash needs through the Loan Agreement with Medytox and the related business combination which may provide such financing that will sustain the Company’s operations as a wholly-owned subsidiary of Medytox until the Company can achieve profitability and positive cash flows.
Until the Company can generate sufficient levels of cash from its operations, the Company expects to continue to finance future cash needs primarily through proceeds from equity financings and collaborative agreements with strategic partners or through a business combination with a company that has such financing in order to be able to sustain its operations until the Company can achieve profitability and positive cash flows. However, the perception that we may not be able to continue as a going concern may cause others to choose not to pursue a business relationship with us due to concerns about our ability to meet our contractual obligations and may adversely affect our ability to raise additional capital.
While CollabRx, Inc. will form the core of our business and operations going forward, we cannot assure you that we will be successful in pursuing our strategic initiative in CollabRx. It is not possible to predict when our business and results of operations will improve. In consideration of these circumstances, the Company may be forced to consider a merger with another company or the liquidation or dissolution of the Company, including through a bankruptcy proceeding. If we were to liquidate or dissolve the Company through or outside of a bankruptcy proceeding, you could lose all of your investment in the Company’s common stock and warrants.
The Company’s investing activities consisted primarily of furniture purchases for additional staff.
Cash Flows
The following table sets forth the primary sources and uses of cash for each of the periods set forth below (in thousands):
|
Year Ended March 31,
|
Six Months Ended
September 30,
|
|||||||||||||||
|
2014*
|
2013*
|
2014**
|
2013**
|
|||||||||||||
|
Cash flows provided by (used in):
|
||||||||||||||||
|
Operating Activities
|
(2,593
|
)
|
(3,838
|
)
|
(1,649
|
)
|
(1,237
|
)
|
||||||||
|
Investing Activities
|
(22
|
)
|
57
|
(17
|
)
|
(11
|
)
|
|||||||||
|
Financing Activities
|
6
|
-
|
1,370
|
-
|
||||||||||||
|
Net decrease in cash and cash equivalents
|
(2,609
|
)
|
(3,781
|
)
|
(296
|
)
|
(1,248
|
)
|
||||||||
* Derived from the Company’s audited financial statements
** Unaudited
Operating Activities: Net cash used in operating activities in all periods presented resulted primarily from our net losses. These losses were adjusted for non-cash charges and changes in components of working capital.
The net cash used in operating activities during fiscal year 2014 was $2,593. The primary changes in our cash flow statement for fiscal year 2014 were due to our net loss of $3,314, partially offset by stock compensation expense, amortization expense, and the recognition of a non-cash loss of foreign exchange differences in the amount $142 from former subsidiaries related to discontinued operations.
The net cash used in operating activities was $3,838 for the year ended March 31, 2013, and consisted primarily of a net loss of $3,928, and stock compensation expense, partially offset by a VAT refund related to the discontinued operations in our former French subsidiary in the amount of 312 Euros.
The net cash used in operating activities during the six months ended September 30, 2014 was $1,649. The primary changes in our cash flow statement for the six months ended September 30, 2014 compared to the corresponding period in the prior fiscal year were due to our net loss of $2,264, partially offset by changes in stock compensation expense, amortization of intangibles, deferred financing expenses of the Company’s recent round of new financing and changes in accounts receivable.
The net cash used in operating activities was $1,237 for the six months ended September 30, 2013. The primary changes in our cash flow statement for the six months ended September 30, 2013 compared to the corresponding period in the prior fiscal year were due our net loss of $1,360 and changes in assets and liabilities of discontinued operations, partially offset by stock compensation expense and amortization of intangibles.
Investing Activities: Net cash (used in) provided by investing activities for the fiscal years ended March 31, 2014 and 2013 was ($22) and $57, respectively. Net cash used in fiscal 2014 was related to the acquisition of property and equipment, primarily computer equipment. Net cash provided by investing activities in fiscal 2013 was primarily consisted primarily of cash received from the CollabRx acquisition, offset by the issuance of a note receivable, also related to the CollabRx acquisition and the purchase of equipment.
For the six months ended September 30, 2014, net cash used in investing activities was $17 and consisted primarily of the purchase of furniture. For the six months ended September 30, 2013, net cash used in investing activities was $11 and consisted of the purchase of the equipment.
Financing Activities: On June 25, 2014, the Company closed an underwritten public offering of 913,500 shares of its common stock, offered at a public offering price of $2.00 per share. The Company netted $1,347 after underwriting expenses of $480. CollabRx used the net proceeds from the offering for general corporate purposes, including development of their products and services, general and administrative expenses and working capital. Aegis Capital Corp. acted as the sole book-running manager for the offering, In addition to the offering of 913,500 shares of common stock, 27,405 warrants to purchase shares of common stock were issued in connection with this offering. These warrants have an exercise price of $2.50 per share and are not exercisable until June 24, 2015 and expire June 24, 2020. Such securities could potentially dilute earnings per share in future periods. Previously deferred financing expenses incurred to raise equity capital related to financing transactions prior to the completion of the financing, as well as other related expenses incurred in the current period, were recognized in the current period and offset the gross proceeds raised.
The Company also received $23 from the sale of 7,101 shares through an at-the-market financing facility through Cantor Fitzgerald in the current fiscal year.
Net cash provided by financing activities totaled $6 in fiscal year 2014. Cash provided in fiscal 2014 was related to the sale of stock from an at market distribution plan (At Market Distribution Plan 2014) as a result of the Company’s filing of an S-3 in its third quarter of fiscal year 2014.
Net cash provided by financing activities for the fiscal year ended March 31, 2013, as well as the six months ended September 30, 2013, respectively, was zero. The Company engaged in no financing activities in these periods.
Operating Capital Requirements
We expect to incur additional operating losses in the near future and our operating expenses will increase as we continue to expand our marketing efforts to drive market adoption of CollabRx products and develop new product offerings. Our liquidity requirements have and will continue to consist of sales and marketing expenses, engineering, research and development expenses, and general corporate expenses.
As CollabRx continues to commercialize its products, we anticipate that our research and development expenditure requirements will also increase in order to expand our product suite and provide access through different technologies. We expect that we will use a portion of the net proceeds of this offering, in combination with our existing cash and cash equivalents, for these purposes. The amount by which we increase our sales and marketing expenses and research and development expenses will be dependent upon the net proceeds of this offering and cannot currently be estimated. We expect that our planned expenditures will be funded from our ongoing operations, as well as from the net proceeds of this offering. We believe that our existing cash and cash equivalents will be adequate to fund the Company’s operations requirements and obligations only through December 31, 2014. We may consider raising additional capital to expand our business, to pursue strategic investments, to take advantage of financing opportunities, or for other reasons. In the future, we expect our operating and capital expenditures to increase as we increase our headcount, expand our marketing and sales activities and continue to invest in new product offerings. As CollabRx revenue grows, we expect our accounts receivable balance to increase. Any increase in accounts payable and accrued expenses may not be completely offset by increases in accounts receivable, which could result in greater working capital requirements.
If our available cash balances, net proceeds from this offering, and anticipated cash flow from operations are insufficient to satisfy our liquidity requirements including because of lower demand for our products or other risks described in this prospectus, we may seek to sell common or preferred equity or convertible debt securities, enter into an additional credit facility or another form of third-party funding, or seek other debt financing. The sale of equity and convertible debt securities may result in dilution to our stockholders and those securities may have rights senior to those of our common shares. If we raise additional funds through the issuance of preferred stock, convertible debt securities or other debt financing, these securities or other debt could contain covenants that would restrict our operations. Any other third-party funding arrangement could require us to relinquish valuable rights. We may require additional capital beyond our currently anticipated amounts. Additional capital may not be available on reasonable terms, or at all.
These estimates are forward-looking statements and involve risks and uncertainties and actual results could vary materially and negatively as a result of a number of factors, including the factors discussed in the section “Risk Factors” of this prospectus. We have based our estimates on assumptions that may prove to be wrong and we could utilize our available capital resources sooner than we currently expect. If we cannot expand our operations or otherwise capitalize on our business opportunities because we lack sufficient capital, our business, financial condition, and results of operations could be materially adversely affected.
Net Operating Loss Carryforwards
The Company analyzes the valuation of its deferred tax assets annually. The deferred tax asset valuation allowance as of March 31, 2014 is attributed to U.S. federal, and state deferred tax assets, which result primarily from future deductible accruals, net operating loss carryforwards, and tax credit carryforwards. We believe that, based on a number of factors, the available objective evidence creates sufficient uncertainty regarding our ability to realize the deferred tax assets such that a full valuation allowance has been recorded. These factors include our history of losses, and the lack of carryback capacity to realize deferred tax assets.
In accordance with Section 382 of the Internal Revenue Code, the amounts of and benefits from net operating loss and tax credit carryforwards may be impaired or limited in certain circumstances. Events which cause limitations in the amount of net operating losses or credits that we may utilize in any one year include, but are not limited to, a cumulative ownership change of more than 50% as defined, over a three year period.
We recognize interest and penalties related to uncertain tax positions in income tax expense. Income tax expense for the year ended March 31, 2014 includes no interest. As of March 31, 2014, we have no accrued interest and penalties related to uncertain tax positions.
Components of loss from continuing operations before income tax benefit is attributed to the following geographic locations for the years ended March 31, 2014 and 2013 (in thousands):
|
Year ended March 31,
|
2014
|
2013
|
||||||
|
Domestic
|
$
|
(3,548
|
)
|
$
|
(4,056
|
)
|
||
|
Foreign
|
-
|
-
|
||||||
|
Loss from continuing operations before income tax benefit
|
$
|
(3,548
|
)
|
$
|
(4,056
|
)
|
||
Components of income tax expense (benefit) for the years ended March 31, 2014 and 2013 consisted of the following (in thousands):
|
Year ended March 31,
|
2014
|
2013
|
||||||
|
Current:
|
||||||||
|
U.S. Federal
|
$
|
-
|
$
|
-
|
||||
|
State and Local
|
2
|
-
|
||||||
|
Foreign (credit)
|
-
|
-
|
||||||
|
Total current tax expense (benefit)
|
2
|
-
|
||||||
|
Deferred
|
||||||||
|
U.S. Federal
|
(81
|
)
|
(83
|
)
|
||||
|
State and Local
|
-
|
-
|
||||||
|
Foreign (credit)
|
-
|
-
|
||||||
|
Total deferred tax expense
|
(81
|
)
|
(83
|
)
|
||||
|
Total income tax expense (benefit)
|
$
|
(79
|
)
|
$
|
(83
|
)
|
||
The income tax expense (benefit) for the years ended March 31, 2014 and 2013 differed from the amounts computed by applying the statutory U.S. federal income tax rate as follows (in thousands):
|
Year ended March 31,
|
2014
|
2013
|
||||||
|
Federal tax expense (benefit) at U.S. Statutory Rate
|
$
|
(1,126
|
)
|
$
|
(1,335
|
)
|
||
|
State tax expense (benefit) net of federal tax effect
|
(193
|
)
|
(246
|
)
|
||||
|
Change in valuation allowance
|
1,196
|
4,572
|
||||||
|
Tax effect of acquired net operating loss carryforwards
|
-
|
(3,123
|
)
|
|||||
|
Foreign Sub Germany
|
251
|
-
|
||||||
|
Amortization of deferred tax liability
|
(81
|
)
|
(83
|
)
|
||||
|
Other items
|
(126
|
)
|
132
|
|||||
|
Total income tax benefit
|
$
|
(79
|
)
|
$
|
(83
|
)
|
||
Components of deferred taxes are as follows (in thousands):
|
Year ended March 31,
|
2014
|
2013
|
||||||
|
Deferred tax assets:
|
||||||||
|
Deferred revenue
|
$
|
48
|
$
|
-
|
||||
|
Accruals, reserves and other
|
1,932
|
1,616
|
||||||
|
Net operating loss carryforwards
|
45,142
|
44,404
|
||||||
|
Credit carryforward
|
2,397
|
2,380
|
||||||
|
Capitalized research and development costs
|
299
|
299
|
||||||
|
Other
|
5
|
9
|
||||||
|
Gross deferred tax assets
|
49,823
|
48,708
|
||||||
|
Valuation allowance
|
(49,823
|
)
|
(48,708
|
)
|
||||
|
Net deferred tax asset
|
$
|
-
|
$
|
-
|
||||
|
Deferred tax liability:
|
||||||||
|
Intangible assets
|
$
|
(500
|
)
|
$
|
(581
|
)
|
||
The Company adopted FASB Interpretation No. 48, “Accounting for Uncertainty in Taxes”, (ASC Topic 740), on January 1, 2007. As a result of the implementation of ASC Topic 740, the Company did not recognize any adjustment to the liability for uncertain tax positions and therefore did not record any adjustment to the beginning balance of accumulated deficit on the consolidated balance sheet. As of the date of adoption, the Company recorded a $1.4 million reduction to deferred tax assets for unrecognized tax benefits, all of which is currently offset by a full valuation allowance and therefore did not record any adjustment to the beginning balance of accumulated deficit on the balance sheet at that time.
|
Tabular Reconciliation of Unrecognized Tax Benefits (in thousands)
|
||||
|
Ending Balance as of March 31, 2012
|
$
|
833
|
||
|
Increase/(Decrease) of unrecognized tax benefits taken in prior years
|
-
|
|||
|
Increase/(Decrease) of unrecognized tax benefits related to current year
|
2
|
|||
|
Increase/(Decrease) of unrecognized tax benefits related to settlements
|
-
|
|||
|
Reductions to unrecognized tax benefits related to lapsing statute of limitations
|
(13
|
)
|
||
|
Ending Balance as of March 31, 2013
|
822
|
|||
|
Increase/(Decrease) of unrecognized tax benefits taken in prior years
|
-
|
|||
|
Increase/(Decrease) of unrecognized tax benefits related to current year
|
77
|
|||
|
Increase/(Decrease) of unrecognized tax benefits related to settlements
|
-
|
|||
|
Reductions to unrecognized tax benefits related to lapsing statute of limitations
|
-
|
|||
|
Ending Balance as of March 31, 2014
|
$
|
899
|
||
There are no positions for which it is reasonably possible that the total amounts of unrecognized tax benefits will significantly increase or decrease within 12 months of the reporting date.
Because the statute of limitations does not expire until after the net operating loss and credit carryforwards are actually used, the statues are still open on fiscal years ended March 31, 1995 forward for federal purposes, and for fiscal years ended March 31, 2003 forward for state purposes. For the years prior to March 31, 2010 for federal purposes and prior to March 31, 2009 for state purposes, any adjustments would be limited to reduction in the net operating loss and credit carryforwards.
Total interest and penalties included in the statement of operations for the year ended March 31, 2014 is zero. It is the Company’s policy to include interest and penalties related to uncertain tax positions in tax expense.
We have recorded no net deferred tax assets for the years ended March 31, 2014 and 2013, respectively. The Company has provided a valuation allowance of $49.8 million and $48.7 million as of March 31, 2014 and 2013, respectively. The valuation allowance fully reserves all net operating loss carryforwards, credits and non-deductible accruals and reserves, for which realization of future benefit is uncertain. The realization of net operating losses may be limited due to change of ownership rules. The valuation allowance increased by $1.1 million in fiscal 2014 and increased by $4.8 million during fiscal 2013.
As of March 31, 2014, the Company has net operating loss carryforwards of approximately $121.5 million and $65.2 million for federal and state tax purposes, respectively. The federal net operating loss carryforward will begin to expire in the year ended March 31, 2020 and the state of California began expiring as of March 31, 2013.
As of March 31, 2014, the Company also has research and experimentation credit carryforwards of $1.4 million and $0.9 million for federal and state income tax purposes, respectively. A portion of the federal credit began to expire in the year ended March 31, 2012 and the state of California will never expire under current law.
The Tax Reform Act of 1986 limits the use of net operating loss and tax credit carryforwards in certain situations where changes occur in the stock ownership of a corporation during a certain time period. In the event the Company had incurred a change in ownership, utilization of the carryforwards could be significantly restricted
Off-Balance Sheet Arrangements
We do not currently have, nor have we ever had, any relationships with unconsolidated entities or financial partnerships, such as entities often referred to as structured finance or special purpose entities, established for the purpose of facilitating off-balance sheet arrangements or other contractually narrow or limited purposes. In addition, we do not engage in trading activities involving non-exchange traded contracts.
Quantitative and Qualitative Market Risk Disclosure
We are exposed to market risks in the ordinary course of our business. Market risk represents the risk of loss that may impact our financial position due to adverse changes in financial market prices and rates. Our market risk exposure is primarily a result of fluctuations in interest rates and foreign currency exchange rates.
Foreign Currency Exchange Risk
As of March 31, 2014 and 2013, all of our investments were classified as cash equivalents in the consolidated balance sheets. Our investment portfolio at fiscal 2014 and fiscal 2013 was comprised of money market funds. With the sale of the DRIE related assets and the closure of the Tegal France subsidiary, our exposure to foreign currency fluctuations has been mostly eliminated. For the fiscal years ended March 31, 2014, and 2013, fluctuations of the U.S. dollar in relation to the Euro were immaterial to our financial statements.
Changes in the exchange rate between the Euro and the U.S. dollar are currently immaterial to our operating results. Exposure to foreign currency exchange rate risk may increase over time as our business evolves. We expect that sales in international markets may account for a significant portion of any future revenue, as the Company plans to market to customers located outside the United States.
Periodically, the Company would enter into foreign exchange contracts to sell Euros, which are used to hedge a sales transaction in which costs were denominated in U.S. dollars and the related revenue was generated in Euros. As of March 31, 2014, there were no outstanding foreign exchange contracts.
Interest Rate Risk
We are only marginally exposed to interest rate risk through interest earned on money market funds. Interest rates that may affect these items in the future will depend on market conditions and may differ from the rates we have experienced in the past. We do not hold or issue derivatives, commodity instruments or other financial instruments for trading purposes.
Critical Accounting Policies and Estimates
Our discussion and analysis of our financial condition and results of operations are based upon our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States of America.
The preparation of these financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses, and related disclosure of contingent assets and liabilities. On an ongoing basis, we evaluate our estimates, including those related to revenue recognition, bad debts, intangible and long lived assets, accrued expenses, and deferred taxes. We base our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
We prepare the condensed consolidated financial statements in conformity with generally accepted accounting principles in the United States (“GAAP”) which requires management to make certain estimates, judgments and assumptions that affect the reported amounts in the accompanying condensed consolidated financial statements, disclosure of contingent assets and liabilities and related footnotes. Accounting and disclosure decisions with respect to material transactions that are subject to significant management judgment or estimates include but are not limited to revenue recognition, accounting for stock-based compensation, accounts for receivables and allowance for doubtful accounts and impairment of long-lived assets. Actual results may differ from these estimates under different assumptions or conditions. Critical accounting policies are defined as those that are required for management to make estimates, judgments and assumptions giving due consideration to materiality, in certain circumstances that affect amounts reported in the condensed, consolidated financial statements, and potentially result in materially different results under different conditions and assumptions. We based these estimates and assumptions on historical experience and evaluate them on an on-going basis to help ensure they remain reasonable under current conditions. Actual results could differ from those estimates. During the twelve months ended March 31, 2014, there were no significant changes to the critical accounting policies and estimates discussed in the Company’s 2014 Annual Report on Form 10-K. During the six months ended September 30, 2014, there were no significant changes to the critical accounting policies and estimates discussed in the Company’s fiscal year 2015 Quarterly Report on Form 10-Q filed on August 14, 2014.
We believe the following critical accounting policies are the most significant to the presentation of our consolidated financial statements:
Revenue Recognition
We derive revenues under multiple arrangements with different customers, depending on specific contractual arrangements, and each contract sale of our interpretive data is evaluated individually in regard to revenue recognition. Our service-based products are sold under an arrangement that requires customers to pay one-time set-up fees, maintenance fees and per-test fees. We recognize the one-time set-up fees upon contract signing which corresponds to the period in which the set-up is performed. The maintenance fees are recognized ratably over the contract period and the per-test fees are recognized as they are incurred and billed. Customers generally enter into one-year renewable annual contracts. However, since most of our existing contracts have been entered into during the past year, we are not yet able to assess the rate at which customers will renew their contracts.
In general, we believe that over time the per-test fees will represent the largest source of revenue in this arrangement. However, since the field is newly emerging and our customers are unable to forecast test volume, it is not possible to determine in advance what proportion of the total revenue may eventually be represented by such per-test fees, so the initial recognized revenues are not a reliable metric for determining the overall value of a customer contract.
We have integrated in our evaluation the related guidance included in Accounting Standards Codification (“ASC”) Topic 605 – “Revenue Recognition”. We recognized revenue when persuasive evidence of an arrangement exists, the seller’s price is fixed or determinable and collectability is reasonably assured. For arrangements that include multiple deliverables, we identify separate units of accounting based on the guidance under ASC 605-25 “Multiple Element Arrangements”, which provides that revenue arrangements with multiple deliverables should be divided into separate units of accounting, if certain criteria are met. The consideration of the arrangement is allocated to the separate units of accounting using the relative selling price method. Applicable revenue recognition criteria are considered separately for each separate unit of accounting. We assess the ability to separate multiple deliverables in accordance with the relevant accounting literature.
Often in the past we have received specific customer requests for additional features and/or content in our products. On such occasions we have charged for “customized” content or services on a fee-for-service basis. These services include the assembly and delivery of portions of our content in discrete lots, customizing user interfaces to specific user requirements, or expansion of our database to include data not previously covered. We expect to continue to offer such fee-for-service contracts in the future, since the field is rapidly changing, and customer requirements are evolving.
Revenue from such contracts is recognized primarily under the percentage of completion method. Under this method we recognize estimated contract revenue and resulting income based on costs incurred to date as a percentage of the total estimated costs as we consider this model to best reflect the economics of these contracts. In such contracts, the Company’s efforts, measured by staff time expended, typically represents the contractual milestones or output measure.
Deferred revenues consist of revenues billed or received that will be recognized in the future under contracts existing at the balance sheet date.
Accounts Receivable – Allowance for Doubtful Accounts
For fiscal years 2014 and 2013, and as of September 30, 2014, the Company had zero reserves for potential credit losses as such risk was determined to be immaterial. The Company does not currently maintain an allowance for doubtful accounts receivable for potential estimated losses resulting from the inability of the Company’s customers to make required payments. The Company believes no such reserve is currently required. The Company had zero write-offs during fiscal years 2014 and 2013 and the three months ended September 30, 2014. The Company reviews the estimated risk of current customers’ inability to make payments on a quarterly basis to determine if any amount is uncollectible.
Fair Value Measurements
We define fair value as the price that would be received from selling an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. When determining fair value measurements for assets and liabilities required or permitted to be recorded at fair value, we consider the principal or most advantageous market in which we would transact and we consider what assumptions market participants would use when pricing the asset or liability, such as inherent risk, transfer restrictions, and risk of nonperformance. The fair value hierarchy distinguishes between (1) market participant assumptions developed based on market data obtained from independent sources (observable inputs) and (2) an entity’s own assumptions about market participant assumptions developed based on the best information available in the circumstances (unobservable inputs). The fair value hierarchy consists of three broad levels, which gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities (Level 1) and the lowest priority to unobservable inputs (Level 3). The three levels of the fair value hierarchy are described below:
|
·
|
Level 1: Quoted prices (unadjusted) in active markets that are accessible at the measurement date for assets or liabilities. |
|
·
|
Level 2: Directly or indirectly observable inputs as of the reporting date through correlation with market data, including quoted prices for similar assets and liabilities in active markets and quoted prices in markets that are not active. Level 2 also includes assets and liabilities that are valued using models or other pricing methodologies that do not require significant judgment since the input assumptions used in the models, such as interest rates and volatility factors, are corroborated by readily observable data from actively quoted markets for substantially the full term of the financial instrument. |
|
·
|
Level 3: Unobservable inputs that are supported by little or no market activity and reflect the use of significant management judgment. These values are generally determined using pricing models for which the assumptions utilize management’s estimates of market participant assumptions. |
In determining fair value, we utilize valuation techniques that maximize the use of observable inputs and minimize the use of unobservable inputs to the extent possible as well as considers counterparty credit risk in its assessment of fair value.
Our financial instruments consist primarily of money market funds. At March 31, 2014, all of our current assets in financial instruments investments were classified as cash equivalents in the consolidated balance sheet. The investment portfolio at March 31, 2014 was comprised of money market funds. Our cash equivalents total $1,430 as of March 31, 2014. The carrying amounts of our cash equivalents are valued using Level 1 inputs. The Company uses the Black-Scholes option pricing model as its method of valuation for warrants that are subject warrant liability accounting. The determination of the fair value as of the reporting date is affected by the Company’s stock price as well as assumptions regarding a number of highly complex and subjective variables which could provide differing variables. These variables include, but are not limited to, expected stock price volatility over the term of the security and risk free interest rate. In addition, the Black-Scholes option pricing model requires the input of an expected life for the securities for which we have estimated based upon the stage of the Company’s development. The fair value of the warrant liability is revalued each balance sheet date utilizing the Black-Scholes option pricing model computations with the decrease or increase in the fair value being reported in the Consolidated Statement of Comprehensive Loss as other income, net. A significant increase (decrease) of any of the subjective variables independent of other changes would result in a correlated increase (decrease) in the liability and an inverse effect on net income. We also have warrant liabilities which are valued using Level 3 inputs.
As of September 30, 2013, the Company’s warrant liability has been extinguished, and the Company has no other financial instruments subject to using Level 3 inputs.
The change in the fair value of warrants is as follows (in thousands):
|
Year Ended March 31,
|
||||||||
|
2014
|
2013
|
|||||||
|
Balance at the beginning of the period
|
$
|
10
|
$
|
19
|
||||
|
Change in fair value recorded in earnings, including expirations
|
(10
|
)
|
(9
|
)
|
||||
|
Balance at the end of the period
|
$
|
-
|
$
|
10
|
||||
Identified Intangible Assets
Intangibles include patents and trademarks that are amortized on a straight-line basis over periods ranging from 3 years to 10 years. We perform an ongoing review of our identified intangible assets to determine if facts and circumstances exist that indicate the useful life is shorter than originally estimated or the carrying amount may not be recoverable. If such facts and circumstances exist, we assess the recoverability of identified intangible assets by comparing the projected undiscounted net cash flow associated with the related asset or group of assets over their remaining lives against their respective carrying amounts. Impairment, if any, is based on the excess of the carrying amount over the fair value of those assets.
Intangible assets, except for trade names, are amortized on a straight-line basis. Intangible assets related to trade names are not amortized. The Company tests for impairment at least annually. The amortization expense included in cost of revenue is related to the acquired software and is amortized on a straight line basis over the expected life of the asset, which the Company believes to be ten years.
No impairment charges for intangible assets were recorded for the fiscal years ended 2014 and 2013. Prior to the acquisition of CollabRx, all of our historical intangible assets, other than those related to NLD and Compact, were included in the asset sale of the DRIE product line to SPTS. NLD is a process technology that bridges the gap between high throughput, non-conformal chemical vapor deposition (“CVD”) and highly conformal, low throughput atomic layer deposition (“ALD”). The portfolio included over 35 U.S. and international patents in the areas of pulsed-CVD, plasma-enhanced ALD, and NLD.
Impairment of Long-Lived Assets
Long-lived assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount may not be recoverable as well as at fiscal year end. If undiscounted expected future cash flows are less than the carrying value of the assets, an impairment loss is recognized based on the excess of the carrying amount over the fair value of the assets. No impairment charges for intangible assets were recorded for the fiscal years ended 2014 and 2013, respectively, since all of our historical intangible assets, other than those related to NLD and Compact, were included in the asset sale of the DRIE product line to SPTS. As our NLD patents and intellectual property were all internally developed (except for those acquired in connection with the Simplus acquisition, which were subsequently written-off) the value of our NLD technology had no recorded value prior to sale.
Long-lived assets also consist of property and equipment. We recorded disposal losses of $0 and $17 for property and equipment for the fiscal years ended March 31, 2014 and 2013, respectively. In fiscal year 2013, we disposed of certain assets in connection with the relocation of our main offices from Petaluma, CA to San Francisco, CA in September 2012.
Deferred Taxes
We record a valuation allowance to reduce our deferred tax assets to the amount that is more likely than not to be realized. Based on the uncertainty of future taxable income, we have fully reserved our deferred tax assets as of March 31, 2014 and 2013. In the event we were to determine that we would be able to realize our deferred tax assets in the future, an adjustment to the deferred tax asset would increase income in the period such determination was made.
Accounting for Stock-Based Compensation
We have adopted several stock plans that provide for issuance of equity instruments to our employees and non-employee directors. Our plans include incentive and non-statutory stock options and restricted stock awards. These equity awards generally vest ratably over a four-year period on the anniversary date of the grant, and stock options expire ten years after the grant date. Certain restricted stock awards may vest on the achievement of specific performance targets. We also have an Employee Stock Purchase Plan (“ESPP”) that allows qualified employees to purchase shares of common stock at 85% of the fair market value on specified dates. The estimates and judgments used in calculating stock-based compensation include the use of expected volatility, forfeiture and interest risk free rates, the expected term of options and the use of the Black-Scholes pricing model. The Company does not pay dividends.
Recent Accounting Pronouncements
In August 2014, the FASB issued ASU 2014-15, Presentation of Financial Statements – Going Concern (Sub Topic 205-40) -Disclosure of Uncertainties about an Entity’s Ability to Continue as a Going Concern. ASU 2014-15 clarifies principles and definitions that may be used by an organization’s management for disclosures that are currently made available in financial statement footnotes. Presently, U.S. GAAP does not provide an organization’s management guidance regarding its responsibility to assess whether substantial doubt exists regarding the ability to continue as a going concern or to prepare related footnote disclosures. Instead, auditors are responsible for assessing an entity’s ability to continue as a going concern under AU-C 570. ASU 2014-15 will move this responsibility to management. ASU 2014-15 will require management to evaluate whether there are conditions or events that raise substantial doubt about the entity’s ability to continue as a going concern for one year from the date the financial statements are issued. ASU 2014-15 is effective for annual periods ending after December 15, 2016 to allow the auditing guidance to catch up with this change. ASU No. 2014-15 affects all companies and nonprofits and early application is allowed.
Overview
Overview of our Current Business
CollabRx, Inc. develops and markets medical information and clinical decision support products and services intended to set a standard for the clinical interpretation of genomics-based, precision medicine. Building on a comprehensive and specialized knowledge base, software systems that facilitate dynamic updating and automated reporting, and a unique network of over 75 independent expert clinical advisors, we have developed a suite of unique and differentiated information products and services. These products include a scalable system for providing high-value information on biomarkers and related therapies for inclusion in reports prepared by diagnostic labs reporting on the results from their multi-biomarker genomic-based tests. In addition, we have developed decision-support tools for physicians and patients to assist with treatment planning for advanced cancer, offered over the Internet in both web-based and mobile apps. We believe that both product sets are highly synergistic, and allow opportunities for enhancement, cross-fertilization and adoption that go beyond what each would offer individually.
Our overall vision is that we are at the dawn of an era of explosive growth of data and information generated at the molecular level that must be interpreted and contextualized into knowledge before it can be used effectively by either physicians or patients. We regard this knowledge as being the most valuable portion of the molecular diagnostic process and we believe that all sectors of the healthcare industry, including providers, insurers, drug developers and patients, are potential users of this knowledge. We aim to deliver our proprietary interpretive content as quickly as possible and in as many usable forms as possible via the Internet.
We are just entering our commercialization phase. The systems and approach that we have developed can be applied to many disease states, but we have chosen to focus initially on cancer, an area of tremendous need and potential. We believe that our approach is unique, disruptive in the market and well timed, offering the potential for high growth, profitability and above average returns for investors.
We support oncologists and pathologists in their efforts to plan treatment approaches for their advanced cancer patients. We do so through three major information products, all of which are based on a proprietary knowledgebase that is informed by external data sources, our in-house experts, and a large network of independent clinical advisors. We attempt to gather together much of the data and evidence that is relevant to different therapeutic approaches in the context of the presence of specific genetic biomarkers, relating those to the relevant drugs and clinical trials that may help a physician plan a treatment approach for a patient. Similar to an electronic library, we focus on keeping the information comprehensive, accurate, current and easy to access. Currently, our information products include the following:
|
Product
|
Users
|
Description
|
Business Model
|
|
Genetic Variant Annotation Service™
(GVA™)
|
Pathologists and Laboratory Medical Directors via cloud-based servers
|
Automated clinical interpretation of tumor genetic alterations (mutation and copy number variation)
|
Laboratories pay $75-$150 per test event or purchase annual subscription
|
|
Therapy Finders™
for Melanoma, Colorectal and Lung Cancer and Metastatic Breast Cancer
|
Oncology professionals at the point-of-care
|
Web-based expert systems for clinical decision support
|
Advertising and sponsorship sharing with on-line media partner MedPage Today
|
|
CancerRx
|
Oncology professionals at the point-of care
|
Mobile app with reference tools, social media, and expert systems
|
Advertising and sponsorship sharing with media partner MedPage Today
|
Since launching our Genetic Variant Annotation Service (GVA) in August 2013, several prominent diagnostic laboratories, comprehensive cancer centers and life science companies have become customers of and/or partners in this product, including: Life Technologies, Inc. (now Thermo-Fisher, Inc., Carlsbad, California), Affymetrix, Inc. (Santa Clara, California), OncoDNA, SA (Brussels, Belgium), Sengenics, Pte., Ltd. (Singapore), Cynvenio Biosystems, Inc. (Westlake Village, California), Quest Diagnostics, Inc. (Madison, New Jersey), CellNetix Pathology and Laboratories (Seattle, Washington), Genoptix (a Novartis company, Carlsbad, California), The Jackson Laboratory for Genomic Medicine (Bar Harbor, Maine), The University of Chicago Medical (Chicago, Illinois), Stanford Hospital Molecular Laboratory (Palo Alto, CA) and The Ohio State University Medical Center (Columbus, Ohio).
Our Therapy Finders™ and CancerRx are made available free to physicians and patients via our on-line media partner, MedPage Today, the professional property of Everyday Health, Inc.
Company Background
CollabRx (f/k/a Tegal) was formed in December 1989 to acquire the operations of the former Tegal Corporation, a division of Motorola, Inc. We designed, manufactured, marketed and serviced specialized systems used primarily in the production of semiconductors and micro-electrical mechanical devices, including integrated circuits, memory devices, sensors, accelerometers and power devices. Beginning in late 2009, we experienced a sharp decline in revenues resulting from the collapse of the semiconductor capital equipment market and the global financial crisis. In a series of transactions from 2010 to 2012, we sold the majority of our operating assets and intellectual property portfolio. During the same time period, our Board of Directors evaluated a number of strategic alternatives, which included the continued operation of the Company as a stand-alone business with a different business plan, a merger with or into another company, a sale of the Company’s remaining assets, and the liquidation or dissolution of the Company. We investigated opportunities within and outside the semiconductor capital equipment industry and evaluated a number of transactions involving other diversified technology-based companies. Throughout this process, we developed and refined our criteria for a business combination, with an eventual focus on the healthcare industry, and specifically information technology and services within the healthcare industry.
On July 12, 2012, we completed the acquisition of a private company called CollabRx, Inc. (the “Merger”), pursuant to an Agreement and Plan of Merger dated as of June 29, 2012 (the “Merger Agreement”). As a result of the Merger, CollabRx became a wholly-owned subsidiary of the Company. In consideration for 100% of the stock of CollabRx, we agreed to issue an aggregate of 236,433 shares of common stock, representing approximately 14% of the Company’s total shares outstanding prior to the closing, to former CollabRx stockholders. As of July 12, 2012, these shares had a fair value of $932. We also assumed $500 of existing CollabRx indebtedness through the issuance of promissory notes. The principal amount of the promissory notes is payable in equal installments on the third, fourth and fifth anniversaries of the closing date of the Merger, along with the accrued but unpaid interest as of such dates. Prior to the closing of the Merger, we provided $300 of bridge financing to CollabRx. After the completion of the Merger, this loan was reclassified to be included as part of the purchase price, and the loan was thereby extinguished. In addition, in connection with the Merger, we granted a total of 368,417 restricted stock units (“RSUs”) and options as “inducement grants” to newly hired management and employees, all subject to four-year vesting and other restrictions. In December 2012, an aggregate of 215,475 RSUs were forfeited in connection with the resignation of James Karis as our Co-Chief Executive Officer.
The Cancer Market and Genomic Testing
Cancer is a worldwide health concern. In 2002, cancer eclipsed heart disease as the number one cause of death in the U.S. for individuals under the age of 85, according to the National Cancer Institute. In the U.S., nearly 12 million individuals are living with a cancer diagnosis, approximately 7 million individuals are being treated for cancer, approximately 1.5 million new cases are diagnosed each year, over 600,000 people die from cancer each year (6 million deaths worldwide), and nearly 1 in 3 females and 1 in 2 males will be diagnosed with cancer in their lifetime, according to the American Cancer Society. Age is the number one risk factor for the development of cancer. Nearly 80% of cancers are diagnosed in individuals aged 55 years and older, which is the fastest growing age segment of the U.S. population according to the U.S. Census Bureau. JPMorgan recently estimated that the total market opportunity for testing in cancer is estimated at $10B in 2014 in the U.S. alone, growing to $25B by 2018, but this opportunity has only been partially tapped.
Cancer treatment and research has been experiencing an unprecedented explosion of knowledge in the past 5-10 years. There are over 500 new cancer therapies in pre-clinical development, thousands of diagnostic labs (private and hospital based), more than 10,000 cancer-related clinical trials are currently ongoing, and over 100,000 cancer-related papers are published in the scientific literature annually, according to PubMed and ClinicalTrials.gov. The knowledge explosion is associated with two interrelated key drivers: (i) cancer is fundamentally a disease of the genome, (i.e., genetic mutations cause cancer), and (ii) the costs and time to sequence genes (and discover mutations that can be targeted for therapy) have been falling at an increasing rate, driven principally by new sequencing technologies commonly referred to as “Next Generation Sequencing” or “NGS”. The concept of a “$1,000 genome” has broad psychological implications since it represents the price point at which leading experts believe that every cancer genome will be sequenced.
Large-scale genetic sequencing studies in tumors are rapidly uncovering mutations in genes that can be selectively targeted by pharmaceutical and biotechnology companies. There were fewer than 10 cancer genes in 2008 in which genetic aberrations were strongly associated with treatment implications. By comparison, there is broad consensus that there are currently approximately 50 such genes based on recent studies, a number that is expected to double in the next year even as additional mutations are being discovered in genes already associated with cancer. Currently there are approximately over 95 biomarkers representing thousands of mutations in aggregate that are associated with at least some level of clinical actionability. This number is expected to increase rapidly as new discoveries are made.
Competitive Strengths
Faced with this explosion of data that results from the sequencing of multiple genes with hundreds of possible mutations within a single patient tumor, hospital laboratories, diagnostic companies and physicians alike are faced with the challenge of learning about evaluating, and staying current with the therapeutic implications of the presence of such mutations in their patient’s tumor biopsies. CollabRx is differentiated and unique as an information company. We are not a diagnostic lab offering a particular test or series of tests in cancer diagnostics. Instead, we have focused exclusively on the information, analysis and interpretation-based steps in the diagnostic workflow, developing and refining the increasingly complex task of delineating the relationship between known or studied biomarkers in cancer with the therapeutic strategies that the published evidence supports. With our web-based and mobile apps, we provide a means for physicians to access our knowledgebase easily at the point-of-care. For laboratories, we provide a credible, third party resource for the dynamic information and analysis that is needed to interpret the results of genetic tests.
In general, the diagnostic testing workflow for multi-gene cancer testing is comprised of the following steps, with CollabRx focused on the last three:
|
·
|
Specimen Handling - including acquisition, transport and acceptance by the lab |
|
·
|
Sample Prep – extraction of DNA from specimen tissue and preparation for NGS testing |
|
·
|
Genetic Analysis – sequencing, which results in raw sequencing data file suitable for exporting from platform |
|
·
|
Bioinformatics – variant calling and quality filtering, resulting in a structured VCF file |
|
·
|
Data Analysis / Interpretation – identification of “actionable” variants and clinical decision support algorithms |
|
·
|
Reporting – visual analysis, report configuration in format suitable for physicians |
|
·
|
Downstream Analytics - compare results across patients; correlate with clinical outcomes; integrate with EMR data |
Our conviction is that the interpretation and reporting of genomic-based test results will become the key differentiator in the market, as opposed to the design and performance of the test itself, given the rapid commoditization of NGS data generation and inherent lack of intellectual property in the sequencing steps. Ultimately, we believe physicians will judge the quality of a diagnostic test based on the quality of the report, and how well it supports the treatment decision process. This requires a fine balance between comprehensiveness of content and brevity, the ability to prioritize test results based on the inclusion of additional test or patient data, methods to explore the supporting evidence, and a variety of means to access the report. All of these features are either in our current products or on our product roadmap and we believe that we have substantially more experience in determining the information that should be included and features of a report than any other company of our type. Furthermore, there are significant capital efficiencies in operating as an information and data analytics company, as opposed to a vertically integrated, clinical laboratory. We believe that the evolving regulatory and competitive landscape in genomics-based medicine favors our approach over that of an integrated lab. We are platform agnostic, independent, adaptable and unregulated.
We believe that diagnostic companies, medical centers, hospital labs and other community-based labs interested in developing a genomics testing capability will confront the challenges associated with developing and maintaining a clinically-oriented, evidence-based biomarker reference database, and increasingly will realize that it is better to “buy” than to “build.” As a first-mover in developing an independently vetted, comprehensive, and frequently updated knowledge base in clinical oncology, as well as the means to address large-scale testing, we believe that we have a significant and sustainable lead over other organizations, including those that have traditionally been involved in or serve the genomics-based research community.
In addition, we believe that it is important to address physician needs for information directly, rather than solely via diagnostic laboratories. For this reason we have continued the development of our web-based and mobile applications, addressing oncologists and pathologists at work and at home, providing a resource for both education and for decision-support. Through these point-of-care products, we strive to build a recognizable brand identity as a reliable and credible resource for molecular information in cancer that extends past “guidelines” or the “standard of care” which are both typically months or years behind where the evidence and thought leaders believe treatment strategies for their advanced cancer patients can be found.
Our ability to compete in these markets and our ability to serve the needs of physicians treating advanced cancer patients rests on a set of principles and ideas that are potentially very disruptive to the markets that we serve and which offer an opportunity for extraordinary growth and profitability. We believe that the following attributes of CollabRx provide a sustainable competitive advantage:
|
·
|
Our proprietary knowledgebase is focused on actionable information for physicians – CollabRx medical and scientific content is organized in a knowledgebase that expresses the relationship between genetic profiles, other aspects of the medical record (e.g., stage, prior treatments), and therapy considerations including molecular diagnostics, medical tests, clinical trials, drugs, biologics, and other information relevant for treatment planning. Capturing how highly respected practicing physicians use this information in the clinical setting further refines the knowledgebase. Importantly, all content is dynamically updated to reflect the continual release of relevant information into the public domain; updates are provided monthly. Our focus is on providing actionable information that physicians can use to plan treatment strategies for their advanced cancer patients and identifying the evidence in the public domain that justifies the therapy options presented. |
|
·
|
Our automated software platform is scalable and capable of handling high test volumes and fast turn-around times – The CollabRx “Semantic Integration Platform” or SIP brings together methods track important changes in molecular oncology from numerous sources, including the published literature and many of the centralized publicly available databases utilized by biomedical and translational clinician/scientists. The SIP is a powerful analytical platform for identifying actionable biomarkers, and incorporates specialized tools that help our knowledge engineers in the curation of the source material. It manages the uploading and analysis of customer provided test results, accumulates and prepares data and reports for export, and provides systems for quality assurance, automated approval, change management, documentation and project management. Our SIP provides CollabRx with a scalable, interactive service that can handle large test volumes and still maintain fast turn-around times for our customers. |
|
·
|
Our large network of independent expert clinical advisors – Over 75 independent, uncompensated expert advisors, organized by both tissue-specific editorial boards and pan-cancer or biomarker-centric boards, provides a unique, unbiased mechanism to inform and prioritize treatment strategies based on evidence. Currently, portions of our knowledge base that inform our Therapy Finders (and which already include markers for histopathology, stage, prior treatment history and molecular tests) are invoked when the biomarker test results also include a diagnosis matching one of our Therapy Finders. Over time, we believe that through the formation of additional editorial boards and the development of additional Therapy Finders, we will have a means to broaden and enrich our knowledge base in a way that addresses what promises to be an evolving need for more complex, comprehensive and independent decision support. We write and publish with our advisors in the peer-reviewed literature and at conference proceedings, select methods and frameworks by which we qualify the clinical actionability of biomarkers, and participate in original studies that leverage these standards. |
|
·
|
Our first-mover advantage and independence - We believe that CollabRx is the first company to have focused exclusively on the information-based, value-added steps of the diagnostic testing workflow in the context of providing clinical grade interpretation of multi-gene testing in cancer, separate from the processing of tissue samples in a laboratory environment. We have built our knowledgebase, software platform and information products over several years with an investment of nearly $20M. Without a diagnostic panel or test of our own, we can remain agnostic both with respect to the test vendor and the testing platform. In addition, since our network of independent expert advisors come from over 26 prominent institutions from 6 countries, we believe that we can also avoid any inherent or institutional bias in the analysis of test results and the formation of therapeutic options for cancer patients. |
Growth Strategy
Our strategic vision is to become a leading source of high value, independently reviewed, actionable information on molecular medicine that supports decision-making by physicians and patients, embodied in tools and services whose performance or use allow us to accumulate data that is valuable to paying segments of the health care ecosystem. We intend to build our information franchise first in oncology, and then expand to other genomics-based disease areas that are amenable to our approach of automated, scalable, and expert-vetted content creation and distribution.
Payors, individual practitioners and patients alike will increasingly want to understand the power and utility of biomarkers and their associated targeted therapies in connection with treatment planning. By making our knowledge base accessible through easy to use web-based and mobile apps, we believe that we can extend our franchise beyond oncology, cross-sell related products and provide a service that is currently unmatched in the health care marketplace.
Our growth strategy includes the following key elements:
Marketing of our Genetic Variant Annotation™ Service (GVA™) into additional segments within the clinical diagnostic laboratory market. Since launching the GVA, we have been engaged primarily in a business development effort with the largest general reference laboratories, with the belief that these laboratories will eventually make up a large portion of all of the genomic tests administered to cancer patients. We have also been successful in attracting some of the most prominent specialty reference laboratories in the US. We intend to continue to formalize our approach to these market segments, relying more on marketing than business development. We have had some success in penetration of the important academic hospital lab segment, but intend to do more in this segment, likely in association with strategic partners. The additional segments of community hospital labs and research labs are not prominent short-term targets for the GVA, but will be addressed via partnerships with other companies for whom these are target markets. While most cancer patients are treated in community hospitals, their in-house laboratories currently rely largely on either the general or specialty reference labs for cancer genomic testing.
Forming strategic partnerships with life science companies to expand GVA biomarker coverage and utility and to engage with them in cooperative marketing efforts. An example of such a strategic partner is Affymetrix, Inc., which has supported our inclusion of Copy Number Variation (CNV) data, which along with sequencing data, provides a comprehensive genetic profile of somatic tumors. In addition to supporting the inclusion of CNV data in our GVA, Affymetrix is supporting a cooperative marketing effort to their clinical research customers. Another life science company with whom we were engaged in the early development of our GVA was Life Technologies, Inc. (now a part of Thermo-Fisher). We are actively seeking additional strategic partnerships with life science and other large companies to both expand the utility of our GVA and to market to our mutual customers.
Forming partnerships with companies in the diagnostic testing workflow to enhance the GVA and to expand our customer base. Particularly in the academic hospital laboratory segment, there is a wide range of capabilities in the area of information technology (IT). A certain base level of IT capability is needed to design, manage and track test results and to prepare reports for ordering physicians. Several companies with whom we have entered agreements, such as GeneInsight, Inc., have developed product offerings in these areas. Our aim is to be able to offer our GVA content through such IT platforms. In addition, we have established a partnership with Omicia, Inc. which offers both IT support to labs and provides a powerful platform and algorithm for genomic research. We are also seeking to tie-up with companies that are integrated into or offer Electronic Medical Records, to facilitate the integration and reporting of additional high value patient data, such as clinical outcomes.
Supporting the advertising and sponsorship sales activities of Everyday Health, Inc. in connection with the current and future Therapy Finders and CancerRx mobile apps. With the successful launch of the CancerRx mobile app in connection with MedPage Today, the sales and marketing teams of Everyday Health have been actively recruiting advertising and sponsorships for the app, which is free to registered users of MedPage Today. We intend to provide our specialized expertise related to the information needs of oncologists and pathologists to promote CancerRx to the largest pharmaceutical and diagnostic companies. In addition, we intend to develop additional Therapy Finders for other cancers and other tools and features that will drive repeat usage for inclusion on both MedPage Today and CancerRx. Eventually, with continued and prolonged use of the app by physicians, we will be able to develop anonymous data sets which we believe will be of assistance to the detailed sales efforts of our sponsors and advertisers.
Recent Developments
On December 6, 2014, we entered into a non-binding letter of intent with Medytox Solutions, Inc. for a potential business combination between the companies. The business combination is subject to, among other things, due diligence, the execution of a definitive agreement, necessary board of director and stockholder approvals and other customary conditions. We cannot assure you that we will enter into a definitive agreement and, if such definitive agreement is entered into, that the contemplated business combination will be consummated.
Pursuant to the Letter of Intent, Medytox Solutions agreed to advance certain funding to CollabRx in contemplation of the business combination. On January 16, 2015, we entered into a Loan and Security Agreement with Medytox Solutions, pursuant to which it is contemplated that Medytox Solutions will loan up to $2,395,644 to CollabRx. We intend to use the proceeds from the Loan and Security Agreement for working capital and general corporate purposes. Amounts borrowed by our company under the Loan and Security Agreement will accrue simple interest at the rate of fifteen percent (15.0%) per annum. As of February 4, 2015, we had borrowed $440,715 under the Loan and Security Agreement. The making of additional advances to our company under the Loan and Security Agreement is completely discretionary on the part of Medytox Solutions. All amounts borrowed under the Loan and Security Agreement mature on December 31, 2015. Upon the occurrence of an event of default under the Loan and Security Agreement, all or a portion of the then outstanding principal and accrued interest under the Loan and Security Agreement is convertible, in the discretion of Medytox Solutions, into shares of our common stock at a conversion price equal to the lower of (i) $0.85 or (ii) the average of the bid and ask prices of ourcommon stock on the trading day immediately prior to the date of conversion; provided, however, that the maximum number of shares issuable to Medytox Solutions is 14.9% of the number of shares of common stock then outstanding. We agreed to secure the payment and performance of our obligations under the Loan and SecurityAgreement by the grant of a security interest in all of our assets.
Also on January 16, 2015, we entered into an Agreement with MedytoxSolutions, pursuant to which we agreed that in the event we enter into a merger or other sale transaction involving at least thirty-five percent (35.0%) of our shares or assets with a party other than MedytoxSolutions, we will pay MedytoxSolutions a $1,000,000 fee. Notwithstanding the foregoing, no fee will be payable to MedytoxSolutions in the event (i) Medytox Solutions has not provided funding to our company of at least $500,000 pursuant to the Loan and Security Agreement or (ii) Medytox Solutions has not funded an advance requested by us under the Loan and Security Agreement, subject to certain exceptions.
Products and Technology
We are focused on developing and delivering content-rich knowledge-based products and services that inform healthcare decision-making, with an emphasis on genomics-based “precision” medicine and big data analytics. Our proprietary content is organized in a knowledge base that expresses the relationship between genetic profiles and therapeutic options, including molecular diagnostics, medical tests, clinical trials, drugs, biologics, and other information relevant for cancer treatment planning. We have developed a method for capturing how practicing physicians use this information in the clinical setting, by incorporating within the knowledge base the views of a large network of independent key opinion leaders in medicine and medical research. We currently deliver our proprietary content to users via web-based applications and services in the “cloud ” serving physicians and their patients in two settings: (i) at the point-of-care in the “clinic”, and (ii) indirectly, as a part of a genetic test report provided to an ordering physician by a diagnostic testing laboratory, (i.e., the “lab”). Portions of our web-based applications are currently available free to physicians and patients through commercial on-line media partners under a license plus advertising or sponsorship revenue sharing arrangement. The content that we offer to laboratories is sold based on a variation of a “Software as a Service” or SaaS business model, in which our content is provided on a one-time, subscription or per test basis. We also receive fee-for-service payments in connection with customized user interfaces to our database.
We use the term “big data” to refer to datasets whose size is beyond the ability of typical database software tools to capture, store, manage, and analyze. We use the term “cloud” to mean a product or service that can be delivered via the Internet, usually on a pay-for-use or subscription basis, versus the purchase and installation of enterprise-based software, which typically requires investments in both software and hardware, often also requiring large-scale customization efforts.
We search publicly available databases as source documents for our knowledgebase. Such databases include those that are available, either free or on a commercial basis, in the areas of clinical trials, drugs, investigational compounds, biomarkers, bioinformatics, cancer oncology and literature. We then aggregate, annotate and integrate these datasets for the purpose of defining the relationship of biomarkers to therapeutic strategies, drugs and clinical trials. None of the individual databases we utilize as sources provide information on the interrelationships of these discrete elements. In addition, CollabRx has developed a process for incorporating the guidance of our network of physician and research advisors in the selection of the most relevant data for specific diagnoses, histopathological data, prior treatments and biomarkers. The result of this software and expert-assisted process is proprietary content that includes decision rules, succinct statements of therapeutic strategy and a comprehensive listing of appropriate drugs and clinical trials, all related to specific aberrations which might be observed in connection with genomic testing.
Although the process and results are proprietary, we always refer to the relevant source documentation that provides the support for the identification of an actionable biomarker, typically a peer-reviewed, published paper. In this way, we avoid the “black-box algorithm problem” which is prevalent in other companies’ predictive analytical models, but is not currently a trusted methodology in medical practice. Our proprietary content is incorporated into our knowledgebase, which is updated regularly with the assistance of a large network of independent advisors, and which forms the basis for all our products and services.
In addition to analyzing the sequencing data that we collect, we intend to pursue collaborative arrangements with other companies and entities that provide contract research services to oncology practices, conduct in-house clinical and translational research, collect information on patient outcomes and link this information to genetic sequencing data, and calculate the relative costs and benefits associated with different diagnostic tests and therapies. We expect such efforts to lead to novel insights and advances to improve the quality of cancer care and reduce the costs of delivering it.
The physicians and researchers within our network of advisors have agreed to participate in our efforts for an indefinite term, on an uncompensated basis, and without a formal agreement. The board assignments, biographies and current affiliations of all of our advisors are posted on our website.
The systems and approach that we have developed for knowledge aggregation, content creation and expert advisory management can be applied to many disease states, but we have chosen to focus initially on genomic medicine in cancer, which is sometimes referred to as “precision oncology.” This is an area of tremendous activity and promise, where clinical research in genomics has given rise to scores of “targeted” therapies that have proven in many cases to prolong the lives of cancer patients. We believe that oncology is also the area of greatest need, where physicians and patients lack convenient access to clear and easy-to-understand information about which drugs, tests and clinical trials should be considered in constructing a cancer treatment plan based on the genetic profile of a tumor. Our overall vision is that we are at the dawn of an era of explosive growth of data and information generated at the molecular level that must be interpreted and contextualized into knowledge before it can be used effectively by either physicians or patients. We regard this knowledge as being the most valuable portion of the molecular diagnostic process and we believe that all sectors of the healthcare industry, including providers, insurers, drug developers and patients are potential users of this knowledge. We aim to deliver our proprietary interpretive content as quickly as possible and in as many usable forms as possible, via the Internet.
Products
Therapy Finders™ and CancerRx Mobile App
Our Therapy Finders™ web-based application serves as an initial user-interface to the underlying knowledge base. It is available free of charge on our website. Our Therapy Finders™ are also offered to registered physicians through MedPage Today, an offering of Everyday Health, Inc. MedPage Today is a rapidly growing online site that serves 96% of all oncologists and 1.6 million monthly online unique users. Our agreement with Everyday Health provides for an annual license fee payable to our Company and sharing of sponsorships and advertising revenue generated by Everyday Health.
Our Therapy Finder™ products are available free-of-charge on our website. Our Therapy Finders are a series of cancer-specific, web-based apps which are accessed by physicians and patients, usually in the physician’s office. After indicating a number of pre-set options related to stage of cancer, histopathology, prior treatments and presence of biomarkers on an input page, the physician is presented with a results page which explains the role of the biomarker, identifies a possible therapeutic strategy for that particular set of inputs, along with tabs associated with searchable lists of relevant drugs and clinical trials. Therapy Finders are an interactive, informational and educational resource for both physicians and patients, and can be used for decision-support. Therapy Finders do not provide the level of detailed information on specific test results that are available from the GVA. Therapy Finders are offered free of charge and the advisors associated with the development and updating of each app are prominently featured. The development and distribution of Therapy Finders is partially supported by sponsorships and advertising revenue. Our aim is to make this tool available as widely as possible, through as many channels as possible, to help community physicians understand the relevance of biomarker testing and the availability of potential therapies for their advanced cancer patients.
Our Therapy Finders™ products are available on both our website and on the MedPage Today website. There is no material difference between the Therapy Finders™ presented on both websites. The only differences are cosmetic, such as background color and placement of individual frames. Initially, the language used in our Therapy Finders presented on our website was geared toward patients. When the opportunity arose in connection with MedPage Today, we modified the language to be more physician-friendly and indicated this by appending “Professional” to the title “Therapy Finder.” In order to avoid confusion, we replaced the patient oriented version on our website with the professional version that is distributed through MedPage Today. At the same time, we also undertook to revise the other Therapy Finders appearing on our website, but not yet distributed through MedPage today, to the professional (physician-friendly) version, and labeled them as such. Nevertheless, we anticipate offering both professional and patient oriented versions of our Therapy Finders in the future.
Recently, we redesigned our Therapy Finders so that they could be accessed by physicians using the iOS operating system from Apple, Inc. via an iPhone or an iPad, and have named this mobile application “CancerRx.” CancerRx was co-developed with MedPage Today of Everyday Health, Inc., with the ownership of the application retained by CollabRx. A special feature of CancerRx is a daily oncology newsfeed from MedPage Today, all with real-time over the air updates. Under our agreement with MedPage Today each company absorbs its own costs for the development, and we share the gross advertising, sponsorship and data analytics revenues associated with the app. We launched CancerRx during the first fiscal quarter of fiscal year 2015.
Genetic Variant Annotation™ Service (GVA™)
Within the clinical laboratory market segment, our current offering provides the clinical interpretation of genetic variants present in human tumor biopsies, and is sold directly to diagnostic labs that perform molecular testing on patients. Our “Genetic Variant Application” or “GVA” is compiled dynamically by our software platform to provide specific insights to a patient’s diagnostic test results on a test-by-test basis. The GVA results are provided to laboratories in a variety of forms, including with a front-end user interface or directly integrated into a customer’s laboratory information management system. Drawing on our interactive and up-to-date knowledge base, a diagnostic lab medical director can select the most relevant insights for a particular patient at the time of testing, and incorporate those insights on potential therapeutic strategies within the report that is transmitted directly back to the ordering physician (typically an oncologist or pathologist). Our content is branded and identified as “Powered by CollabRx” within the test report. We deliver the GVA product via a cloud-based variation on a SaaS business model, and we receive payment directly from the diagnostic laboratory, typically on a per-test basis. Because we are independent and focused exclusively on providing information on actionable biomarkers, we are able to offer our service to many of the hundreds of laboratories globally that offer genetic testing of cancer tumors.
The GVA is a service offered to diagnostic testing laboratories, including academic medical centers and commercial laboratories. Our lab customers provide us with a test result, usually in the form of an electronic file that represents the results of a genomic test, typically from a “Next Generation Sequencing” (NGS) or similar testing platform. We analyze the test results for the purpose of identifying those aberrations which we have annotated in advance as being “actionable” (i.e., related to a therapeutic strategy). We provide the testing lab with a report, incorporating specific information regarding identified biomarkers and associated therapeutic strategies for each, along with relevant drugs and clinical trials, to a level and in a format that we have agreed in advance with our customer. We are compensated for this service either on a per-test or on a volume-adjusted subscription basis. This service is not available to the public and is not available on our website. To date we have signed SaaS-based, multi-year agreements with Life Technologies, Inc. (Carlsbad, California), OncoDNA, SA (Brussels, Belgium), Sengenics, Pte., Ltd. (Singapore), Cynvenio Biosystems, Inc. (Westlake Village, California), Quest Diagnostics, Inc. (Madison, New Jersey), CellNetix Pathology and Laboratories (Seattle, Washington), Genoptix (a Novartis company)(Carlsbad, California), The Jackson Laboratory for Genomic Medicine (Bar Harbor, Maine).
Technologies
The knowledge base that underlies our clinical and laboratory is focused on the “actionable” molecular biomarkers and evidence-based medicine that guides the selection of therapeutic options. We determine “actionability” based on a defined set of measures of the strength of evidence and other objective criteria supporting different levels of “actionability”. The information that we aggregate, synthesize and report to physicians is based solely on data available publicly in the medical literature. It is referenced with respect to its source documentation and is vetted for appropriateness and relevance as needed by our network of more than 75 independent key opinion leaders, whose identities and biographies are posted on our website. In these important ways we are transparent in our approach to providing the information that supports the day-to-day decisions made by practicing physicians. We have simplified and made more efficient the process by which many physicians would otherwise collect the needed information to make or support clinical decisions (e.g., web search followed by reading). We have performed the searches and compiled the relevant information in advance on behalf of users, ensuring that the information is comprehensive, relevant and up-to-date. Basically, we provide an easy-to-use, efficient, interactive on-line library for practicing oncologists and laboratory medical directors.
We have developed sophisticated, artificial-intelligence-based software programs that allow us to aggregate data from publicly available sources of published, peer-reviewed scientific and medical literature, abstracts and case reports. Our “Semantic Integration Platform”, or SIP, allows us to update on a regular and frequent basis a proprietary knowledge base that links several external and internal databases with information on known and emergent biomarkers, molecular tests that are available to assist with further diagnoses, drugs and compounds that have either been approved as drugs or are under investigation, and the relevant clinical trials that are recruiting patients for further research. All of this information is referenced to published source documentation. We annotate and curate the basic information, creating high-level summaries designed to contextualize for physicians and patients the relationships between the identified biomarkers and the available testing and treatment options.
Fundamental to our business is the concept that “thought-leader” medicine drives advances in clinical practice. Physicians and researchers in the major cancer centers in the United States and abroad that oversee cutting-edge clinical research are discovering new treatment and testing options for patients at an increasingly rapid pace, due in large part to advances in testing and information technology. Treatment options that are incorporated into routine clinical practice “standard of care” guidelines fail to keep up with the rapid pace of discovery in the research laboratories. We have addressed this problem by assembling a network of over 75 leading oncologists and researchers and by providing them with a platform to integrate their knowledge into clinical practice and to distribute that knowledge widely to other practicing physicians. Generally speaking, most patients at this stage are “beyond the standard of care.” We believe this “democratization” of thought-leader medicine is disruptive to the status-quo of compartmentalized, institution-based diagnosis and treatment.
Building on the well-established conceptual framework for publishing in medicine, we have assembled a network of Editorial and Advisory Boards of independent physicians and researchers, based around specific expertise in organ or location-based cancers (e.g., melanoma, colorectal, breast, prostate, etc.) and “pan-cancer” (a biomarker-centric, non-location specific view). Each Editorial Board has a Chairperson and consists of 6 – 12 additional experts recruited by the Chair and assembled specifically to help us model each disease on a molecular level, to create decision nodes for the consideration of additional testing or therapy options, and to weigh alternative treatments against the highest quality of peer-reviewed scientific and medical evidence. Several of our models have been co-authored by our Editorial Board members and published in open access, peer-reviewed journals. The decision-support features of the knowledge have been developed into easy-to-use, web-based Therapy Finder™ applications that we have made available to physicians and patients free of charge on our website and through other online media outlets. In this way, we fulfill our commitment to transparency and the democratization of thought-leader medicine.
Customers
As we transitioned into healthcare, our customers changed materially. Until February 9, 2011, our sales were primarily to large semiconductor and micro-electrical mechanical systems (“MEMS”) device manufacturers. In fiscal year 2013, three customers accounted for 100% of our revenues. Specifically, two of our customers, Life Technologies, Inc. and Everyday Health Inc. accounted for 75% of our revenues, and our management contract with Sequel Power accounted for 25% of our revenues. In fiscal year 2014, five customers accounted 96% of our revenues and one of our customers, Life Technologies, Inc., accounted for 76% of our revenues. No other customer accounted for 10% or more of our revenues in fiscal years 2013 or 2014. Our management contract with Sequel Power was terminated on March 31, 2013, and we have received no revenue from that source since that time. As we continue to make inroads into the commercialization phase of our current business, we expect that our customer base will expand and that our sales will be less concentrated. As of March 31, 2014, four customers accounted for 100% of our accounts receivable balance. One customer accounted for 100% of our accounts receivable balance as of March 31, 2013.
For the three months ended September 30, 2014, three customers accounted for 28.4%, 28.4% and 14.2%, respectively, of the Company’s revenue. For the six months ended September 30, 2014, four customers accounted for 20.9%, 20.9%, 17.4% and 10.4%, respectively, of the Company’s revenue. For the three and six months ended September 30, 2013, one customer accounted for 99.6% and 96.0%, respectively, of the Company’s revenue.
Life Technologies, Inc. has been a major contributor to our revenues and gross profits for the past two years. However we have funded the Company’s operating expenses primarily with cash on hand and the net proceeds from the sale of discontinued assets. Please see the “Liquidity and Capital Resources” section set forth in Item 2 herein. We are actively engaged in negotiations with several other companies who are interested in purchasing our content on similar terms or under annual subscriptions or SaaS arrangements.
Marketing, Sales and Service
We focus on content creation through the aggregation of peer-reviewed published data and its review and interpretation by clinical experts, and the incorporation of that content into products that provide current, credible and actionable information to users. Updated frequently, such information is highly valuable to several segments of the healthcare market, including patients, healthcare providers (e.g., oncologists and pathologists), provider networks, laboratories, diagnostic companies, medical institutions, pharmaceutical and biotechnology companies, and contract research organizations. The diversity of potential users of such information requires a corresponding diversity in marketing approaches and sales strategies. For this reason, we have chosen to enter the markets through strategic partnering arrangements with companies that already have a significant presence in each of the market segments.
For our clinical products, we formed our first strategic partnership with Everyday Health, Inc. a leading on-line media company in the healthcare market. Our agreement with Everyday Health includes license fees and advertising revenue sharing in connection with making our CollabRx Therapy Finders™ available to registered physicians through MedPage Today, Everyday Health, Inc.’s rapidly growing online site that serves 96% of all oncologists and has 1.6 million monthly online unique users.
For our laboratory products, we entered into a multi-year agreements with several companies, including Life Technologies, Inc., Quest Diagnostics, Inc., Affymetrix, Inc., The Jackson Laboratory, CellNetix, Inc., Genoptix, Inc., Cynvenio Biosystems, Inc., OncoDNA, SA (Brussels, Belgium), and Sengenics, Pte., Ltd. (Singapore).
We are in the process of pursuing and negotiating strategic partnerships with other companies in the major healthcare segments as part of a broad business development strategy in which several of our employees, including our senior executives, are involved. Our other marketing efforts consist primarily of our website and presentations by our executives at industry trade shows and conferences. At the present time, we do not engage in direct sales activities to users, and our service activities are limited to supporting and maintaining our software applications that run on several cloud-based servers.
Intellectual Property
Our business relies, in part, upon our ability to protect our proprietary technologies, methods and processes, product designs and branding that we have invented, developed or licensed. To accomplish these objectives, we rely on a combination of patent, trademark, copyright and trade secret laws in the United States and other jurisdictions, as well as license agreements and other contractual protections. Our policy is to seek patent protection and trademark registration for commercially valuable assets we develop, as appropriate, and maintain as trade secrets other aspects of our proprietary platform, processes, and know-how.
As of September 30, 2014, we have licensed the rights to two U.S. patent applications to GeneKey, Inc. pursuant to royalty agreements and have filed one provisional U.S. patent application. We also rely on several registered and unregistered trademarks to protect our brand. In addition, we seek to protect our intellectual property rights by generally requiring our employees and independent contractors involved in development to enter into agreements acknowledging that all inventions, trade secrets, works of authorship, developments, concepts, processes, improvements and other works generated by them on our behalf are our property, and assigning to us any rights, including intellectual property rights, that they may claim in those works.
Despite our efforts to protect our proprietary technologies and our intellectual property rights, unauthorized parties may attempt to copy aspects of our products or obtain and use our trade secrets or other confidential information. We generally enter into confidentiality agreements with our employees, consultants, vendors and customers, and generally limit access to and distribution of our confidential information and proprietary technology. These agreements may not effectively prevent unauthorized use or disclosure of our intellectual property or technology and may not provide an adequate remedy in the event of unauthorized use or disclosure of our intellectual property or technology. In addition, others may independently discover our trade secrets and confidential information, and in such cases we could not assert any trade secret rights against such parties. We cannot assure you that the steps taken by us will prevent misappropriation of our trade secrets or technology. In addition, the laws of some foreign countries do not protect our intellectual property rights to as great an extent as the laws of the United States, and many foreign countries do not enforce these laws as diligently as government agencies and private parties in the United States.
Costly and time-consuming litigation could be necessary to enforce and determine the scope of our trade secret rights and related confidentiality and nondisclosure provisions, and failure to obtain or maintain trade secret protection, or our competitors’ obtainment of our trade secrets or independent development of unpatented technology similar to ours or competing technologies, could adversely affect our competitive business position.
Litigation or proceedings before the U.S. Patent and Trademark Office, or USPTO, or other governmental authorities and administrative bodies in the United States and abroad may be necessary in the future to enforce our intellectual property rights, to protect our patent rights, trademarks, and trade secrets and to determine the validity and scope of the intellectual property rights of others. Our efforts to enforce or protect our intellectual property rights may be ineffective and could result in substantial costs and diversion of resources and management time, and could substantially harm our results of operations.
Competition
Competition in the “content” space can originate from the cancer Internet, online medical journals, consumer-facing healthcare websites, other proprietary databases, and subscription-based services. However, we believe that none of the existing competitors offer the array of experts, vetted content, tools and services that are embodied in the CollabRx organization.
Competition in the clinical decision support space comes primarily from clinical treatment guidelines publishers (e.g., NCCN), boutique-level consulting companies (e.g., N-of-One, Inc.), companies that develop healthcare applications (“apps”, e.g., Athena Health/Epocrates), and more recently laboratories that conduct genomic testing (e.g., Foundation Medicine and Caris Life Sciences). The most relevant direct competitor to CollabRx interpretive analytics and test reporting services is Foundation Medicine, currently recognized as one of a small number of lab testing companies that also provide “best-in-class” interpretive reporting of tumor mutational profiling. CollabRx has been identified as a key potential competitor to Foundation Medicine and others since we broadly enable others to meet and exceed the standard set by Foundation Medicine with respect to test report quality.
Competition in the “analytics” space comes primarily from large firms with a broad focus (e.g., SAP) and from niche firms with a focus in healthcare (e.g., GNS Healthcare) or cancer genomics (e.g., Molecular Health). Both types of firms currently develop and apply statistical models to identify trends in large and complex datasets, but do not routinely provide a clinically relevant interpretive framework to the results. When they do, it’s typically in the content of drug toxicity, and not efficacy. At present these firms represent potential CollabRx partners, but could conceivably become direct competitors if they developed a clinical expert-backed content strategy similar to CollabRx. In addition, unlike CollabRx, these types of firms (i.e., ones that utilize statistical modeling and algorithms) are likely to face much more intense regulatory oversight and scrutiny, based on recent guidance issued by the FDA.
Governmental Regulations
FDA
The United States Food and Drug Administration, or “FDA”, regulates the sale and distribution in interstate commerce of medical devices under the Federal Food, Drug, and Cosmetic Act, or the “FDCA”, including in vitro diagnostic devices, reagents and instruments used to perform diagnostic testing. As CollabRx does not conduct laboratory developed tests (“LDTs”), nor produce or distribute any product that can be categorized as “devices” by the FDA, we do not believe that we are subject to FDA oversight.
HIPAA and HITECH
Under the administrative simplification provisions of the Health Insurance Portability and Accountability Act, or “HIPAA”, as amended by the Health Information Technology for Economic and Clinical Health Act, or “HITECH”, the United States Department of Health and Human Services issued regulations that establish uniform standards governing the conduct of certain electronic health care transactions and protecting the privacy and security of protected health information used or disclosed by health care providers and other covered entities. Three principal regulations with which we are required to comply have been issued in final form under HIPAA: privacy regulations, security regulations, and standards for electronic transactions, which establish standards for common health care transactions. The privacy regulations cover the use and disclosure of protected health information by health care providers. They also set forth certain rights that an individual has with respect to his or her protected health information maintained by a health care provider, including the right to access or amend certain records containing protected health information or to request restrictions in the use or disclosure of protected health information. The security regulations establish requirements for safeguarding the confidentiality, integrity, and availability of protected health information that is electronically transmitted or electronically stored. The HITECH Act, among other things, established certain health information security breach notification requirements. A covered entity must notify any individual whose protected health information is breached. The HIPAA privacy and security regulations establish a uniform federal “floor” and do not supersede state laws that are more stringent or provide individuals with greater rights with respect to the privacy or security of, and access to, their records containing protected health information. Massachusetts, for example, has a state law that protects the privacy of personal information of Massachusetts residents.
These laws contain significant fines and other penalties for wrongful use or disclosure of protected health information. Additionally, to the extent that we submit electronic health care claims and payment transactions that do not comply with the electronic data transmission standards established under HIPAA and HITECH, payments to us may be delayed or denied.
Federal, State and Foreign Fraud and Abuse Laws
In the United States, there are various fraud and abuse laws with which we must comply and we are potentially subject to regulation by various federal, state and local authorities, including CMS, other divisions of the U.S. Department of Health and Human Services (e.g., the Office of Inspector General), the U.S. Department of Justice and individual U.S. Attorney offices within the Department of Justice, and state and local governments. We also may be subject to foreign fraud and abuse laws.
In the United States, the federal Anti-Kickback Statute prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving remuneration to induce or in return for patient referrals for, or purchasing, leasing, ordering or arranging for the purchase, lease or order of, any health care item or service reimbursable under a governmental payor program. Courts have stated that a financial arrangement may violate the Anti-Kickback Statute if any one purpose of the arrangement is to encourage patient referrals or other federal health care program business, regardless of whether there are other legitimate purposes for the arrangement. The definition of “remuneration” has been broadly interpreted to include anything of value, including gifts, discounts, credit arrangements, payments of cash, consulting fees, waivers of co-payments, ownership interests and providing anything at less than its fair market value. Recognizing that the Anti-Kickback Statute is broad and may technically prohibit many innocuous or beneficial arrangements within the health care industry, the U.S. Department of Health and Human Services issued a series of regulatory “safe harbors.” These safe harbor regulations set forth certain provisions, which, if met, will assure health care providers and other parties that they will not be prosecuted under the federal Anti- Kickback Statute. Although full compliance with these provisions ensures against prosecution under the federal Anti-Kickback Statute, the failure of a transaction or arrangement to fit within a specific safe harbor does not necessarily mean that the transaction or arrangement is illegal or that prosecution under the federal Anti-Kickback Statute will be pursued. Penalties for federal anti-kickback violations are severe, and include imprisonment, criminal fines, civil money penalties, and exclusion from participation in federal health care programs. Many states also have anti-kickback statutes, some of which may apply to items or services reimbursed by any third-party payor, including commercial insurers.
Legislation defining two new federal crimes related to health care were recently enacted: health care fraud and false statements relating to health care matters. The health care fraud statute prohibits knowingly and willfully executing a scheme to defraud any health care benefit program, including private payors. A violation of this statute is a felony and may result in fines, imprisonment or exclusion from governmental payor programs such as the Medicare and Medicaid programs. The false statements statute prohibits knowingly and willfully falsifying, concealing or covering up a material act or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for health care benefits, items or services. A violation of this statute is a felony and may result in fines, imprisonment or exclusion from governmental payor programs.
Finally, another development affecting the health care industry is the increased enforcement of the federal False Claims Act and, in particular, actions brought pursuant to the False Claims Act’s “whistleblower” or “qui tam” provisions. The False Claims Act imposes liability on any person or entity that, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment by a federal governmental payor program. The qui tam provisions of the False Claims Act allow a private individual to bring actions on behalf of the federal government alleging that the defendant has defrauded the federal government by submitting a false claim to the federal government and permit such individuals to share in any amounts paid by the entity to the government in fines or settlement. When an entity is determined to have violated the False Claims Act, it may be required to pay up to three times the actual damages sustained by the government, plus civil penalties ranging from $5,500 to $11,000 for each false claim.
In addition, various states have enacted false claim laws analogous to the federal False Claims Act, although many of these state laws apply where a claim is submitted to any third-party payor and not merely a governmental payor program.
In Europe various countries have adopted anti-bribery laws providing for severe consequences, in the form of criminal penalties and/or significant fines, for individuals and/or companies committing a bribery offence. Violations of these anti-bribery laws, or allegations of such violations, could have a negative impact on our business, results of operations and reputation. For instance, in the United Kingdom, under the new Bribery Act 2010, which went into effect in July 2011, a bribery occurs when a person offers, gives or promises to give a financial or other advantage to induce or reward another individual to improperly perform certain functions or activities, including any function of a public nature. Bribery of foreign public officials also falls within the scope of the Bribery Act 2010. Under the new regime, an individual found in violation of the Bribery Act of 2010, faces imprisonment of up to 10 years. In addition, the individual can be subject to an unlimited fine, as can commercial organizations for failure to prevent bribery.
Physician Referral Prohibitions
Under a federal law directed at “self-referral,” commonly known as the “Stark Law,” there are prohibitions, with certain exceptions, on referrals for certain designated health services, including laboratory services, that are covered by the Medicare and Medicaid programs by physicians who personally, or through a family member, have an investment or ownership interest in, or a compensation arrangement with, an entity performing the tests. The prohibition also extends to payment for any testing referred in violation of the Stark Law. A person who engages in a scheme to circumvent the Stark Law’s referral prohibition may be fined up to $100,000 for each such arrangement or scheme. In addition, any person who presents or causes to be presented a claim to the Medicare or Medicaid programs in violation of the Stark Law is subject to civil monetary penalties of up to $15,000 per bill submission, an assessment of up to three times the amount claimed and possible exclusion from participation in federal governmental payor programs. Bills submitted in violation of the Stark Law may not be paid by Medicare or Medicaid, and any person collecting any amounts with respect to any such prohibited bill is obligated to refund such amounts. Many states have comparable laws that are not limited to Medicare and Medicaid referrals.
Corporate Practice of Medicine
Numerous states have enacted laws prohibiting business corporations, such as us, from practicing medicine and employing or engaging physicians to practice medicine, generally referred to as the prohibition against the corporate practice of medicine. These laws are designed to prevent interference in the medical decision-making process by anyone who is not a licensed physician. For example, California’s Medical Board has indicated that determining what diagnostic tests are appropriate for a particular condition and taking responsibility for the ultimate overall care of the patient, including providing treatment options available to the patient, would constitute the unlicensed practice of medicine if performed by an unlicensed person. Violation of these corporate practices of medicine laws may result in civil or criminal fines, as well as sanctions imposed against us and/or the professional through licensure proceedings. Typically such laws are only applicable to entities that have a physical presence in the state.
Segment and Geographical Information
We operate in one reportable business segment and currently derive revenue from the United States alone, although we have signed SaaS-based, multi-year agreements with OncoDNA, SA (Brussels, Belgium) and Sengenics, Pte., Ltd. (Singapore).
Employees
As of September 30, 2014 we had 14 full-time employees. Of our regular employees, eight are in engineering/research and development, and three are in executive and administrative positions. Of the fourteen regular employees, eight hold advanced degrees, including PhDs, MDs and MBAs.
None of our employees is represented by a labor union or covered by a collective bargaining agreement.
Facilities
Our headquarters, encompassing our executive office and storage areas, is located in San Francisco, California. We have a primary lease for office space, consisting of 2,614 square feet, which expires August 31, 2017. We also rent storage/workspace areas on a monthly basis. Other than the equipment lease for copiers, we own all of the equipment used in our facilities. Such equipment consists primarily of computer related assets. We believe these facilities are sufficient to meet our current needs.
Research and Development
Our research and development, or “R&D”, efforts span a broad range of activities, including research into peer-reviewed published literature and databases, the development and publication of Molecular Disease Models, or MDMs, the creation of proprietary knowledge bases of medical and scientific content, the development of applications and user interfaces to access the knowledge bases, and the development of a suite of artificial intelligence-based tools that assist in the research, aggregation, organization, curating and updating of the knowledge bases.
We employ approximately eight full-time scientists and engineers in our R&D organization. The expenses related to R&D resulted from the change in categorization of certain employee related expenses from Engineering to R&D. The efforts of our engineering group include supporting existing product offerings as well as developing future product offerings. We have segregated these expenses and reported expenses that make up the total R&D expenses for respective the fiscal periods.
Research and development expenses for continuing operations for the three and six months ended September 30, 2014 and the years ended March 31, 2014 and 2013 were $32, $82, $284 and $536, respectively. We include all of those employees who work both on engineering activities and R&D activities in the headcount within Engineering and allocate the expense to R&D, as categorized above. The efforts of the engineering group include supporting existing product offerings as well as developing future product offerings. Consequently, such expenses are segregated. The decrease in R&D expenses of $252 in fiscal year 2014 compared to fiscal year 2013 is related to the efforts of Engineering being directed to supporting new customer offerings.
Since R&D is an essential part of our business, we expect that our absolute spending will remain at current levels or increase in the future.
Legal Proceedings
As of September 30, 2014, we had no pending material legal proceedings. From time to time, we may become involved in legal proceedings in the normal course of business and do not expect them to have a material adverse effect on our business.
Directors and Executive Officers
Our Board of Directors and their respective ages and positions:
|
Name
|
Age
|
Director
Since
|
|
Paul Billings, Director
|
62
|
2014
|
|
James Karis, Director
|
66
|
2012
|
|
Jeffrey M. Krauss, Director
|
58
|
1992
|
|
Thomas R. Mika, President & CEO, Acting Chief Financial Officer, Secretary and Treasurer, Chairman of the Board of Directors
|
63
|
2002
|
|
Carl Muscari, Director
|
63
|
2007
|
Paul R. Billings, MD, PhD, FACP, FACMGjoined our Board of Directors on April 7, 2014. Dr. Billings, is a nationally recognized expert on genomic and precision medicine and a board certified internist and clinical geneticist whose career has been devoted to improving patient care by expanding the use of medically relevant genomic technologies in clinical settings, most recently as Chief Medical Officer of Life Technologies, Inc. (acquired by Thermo Fisher Scientific, Inc. Scientific, Inc. in March, 2014). Currently, Dr. Billings serves in multiple roles in industry and government, including as Executive Chairman, Melanoma Diagnostics, Inc., and director of Trovagene, Inc., DecisionQ, Inc. and PAX Neuroscience, Inc. He was Co-Founder and first Medical Director of the Cordblood Registry, Inc., past Senior Physician and SVP of Laboratory Corporation of America, Inc. (LabCorp), Co-Founder and Past Director of Omicia, Inc., Founder and EVP of GeneSage, Inc., and past Director of Ancestry.com, Inc. Dr. Billings currently serves on the Scientific Advisory Board of the FDA, the Genomic Medicine Advisory Committee at the Dept. of Veterans Affairs, and the National Academy of Sciences Institute of Medicine’s Roundtable on Genomics. Dr. Billings was the former Director and Chief Science Officer of the Genomic Medicine Institute at El Camino Hospital. He was also a member of the United States Dept. of Health and Human Services Secretary’s Advisory Committee on Genetics, Health and Society. Dr. Billings has held academic appointments at prestigious universities including Harvard University, UCSF, Stanford University and UC Berkeley, and has served as a physician at numerous medical centers. He is the author of nearly 200 publications and books on experimental and clinical medicine. His work on genetic discrimination was instrumental in the creation and passage of the federal Genetic Information Non-Discrimination Act of 2008. Dr. Billings holds an M.D. from Harvard Medical School and a Ph.D. in immunology from Harvard University.
The Board of Directors has determined that Dr. Billings’ substantial work experience in companies in the management and healthcare industry and his education give him the appropriate set of skills to serve as a member of our Board of Directors.
James M. Karis joined our Board of Directors in July 2012 with our acquisition of CollabRx where he served as its Chief Executive Officer from September 2011. Mr. Karis served as Co-Chief Executive Officer of our company from July 2012 until December 2012, after which he was appointed as CEO of Mapi Group, a service provider to the global healthcare industry. Prior to CollabRx, Mr. Karis was an independent consultant from May 2009 to September 2011. From January 2000 until May 2009, he served as President and Chief Executive Officer of Entelos, Inc., a U.S. based life sciences technology company. Prior to Entelos, he held senior positions in the contract research industry, serving as Chief Operating Officer and President of PAREXEL International Corporation, and earlier, as Chief Operating Officer of Pharmaco International. He was the Vice President of International Operations for Baxter International and a founder of KMR Group, a leading pharmaceutical R&D benchmarking consulting firm. Mr. Karis serves on the Board of Directors of Datatrak, Inc. and also for one private company. Mr. Karis holds a Bachelor of Science degree in Management and Economics from Purdue University and a Masters degree in Applied Economics from The American University.
The Board of Directors has determined that Mr. Karis’ substantial work experience in companies in the management and healthcare industry and his education give him the appropriate set of skills to serve as a member of our Board of Directors.
Jeffrey M. Krauss has served as a director of our company since June 1992. Since February 2012, Mr. Krauss has been a managing member of PPC Enterprises, LLC a private equity firm, where he heads the firm’s healthcare practice. From April 2000 until February 2012 Mr. Krauss was a Managing Member of Psilos Group Managers, LLC, a New York based venture capital firm, and a Managing Member of the general partner of Psilos Group Partners I, LP, Psilos Group Partners II, LP, Psilos Group Partners II-S, LP and Psilos III, each a venture capital partnership. From 1990 until April 2000, Mr. Krauss was a general partner of the general partner of Nazem & Company III, L.P. and Nazem & Company IV, L.P., both venture capital funds. He was also a general partner of The Transatlantic Fund, a joint venture capital fund between Nazem & Company and Banque Nationale de Paris of France. Prior to joining Nazem & Company, Mr. Krauss was a corporate attorney with the law firm of Simpson Thacher & Bartlett, where he specialized in leveraged buyout transactions. He currently serves as a director of several private companies.
Due to Mr. Krauss’ long history with our company and his extensive experience as an investor in various companies, the Board of Directors believes that Mr. Krauss has skills enabling him to contribute meaningfully to our Board of Directors and CollabRx.
Thomas R. Mika was appointed our President and Chief Executive Officer in March 2005 and appointed Chairman of the Board in October 2006. Mr. Mika has more than 25 years of senior management, finance and consulting experience. Serving on our Board of Directors for ten years from 1992 to 2002, which included periods of service as the Chairman of the Compensation Committee and a member of the Audit Committee, until he was appointed as Executive Vice President and Chief Financial Officer in August 2002. Mr. Mika played a key role in Company management, including managing the activities leading to our initial public offering in 1995. Prior to becoming our Executive Vice President and Chief Financial Officer, Mr. Mika founded IMTEC, a boutique investment firm active in the management of several companies. Mr. Mika was also a director of Metrologix, a semiconductor metrology company, from the time of its initial start-up until its sale to KLA-Tencor Corp. Prior to forming IMTEC, Mr. Mika was a managing consultant with Cresap, McCormick & Paget and a policy analyst for the National Science Foundation. He holds a Bachelor of Science degree in microbiology from the University of Illinois at Urbana-Champaign and a Master of Business Administration degree from the Harvard Graduate School of Business.
The Board of Directors has concluded that Mr. Mika should serve on CollabRx’s Board of Directors based on his deep knowledge of CollabRx gained from his positions as President and Chief Executive Officer, as well as his substantial senior management, finance and consulting experience.
Carl Muscari has served as a director of our company since November 2007. Mr. Muscari is currently the Chief Executive Officer of MSRC Co., a leading independent distributor of computer and electronics components based in Brentwood, New Hampshire. During his tenure at MSRC, Mr. Muscari has been credited with the turn-around and modernization of this privately held company. From 1999 until 2003, Mr. Muscari served as Chairman and CEO of Video Network Communications, Inc., based in Portsmouth, New Hampshire. Prior to VNCI, Mr. Muscari was President of Acuity Imaging, Inc., a machine vision company, and President & CEO of Exos, Inc. a private company with force-feedback controls technology incorporated into home video, arcades and PCs, which was sold to Microsoft in 1996. He was Executive Vice President and Chief Operating Officer of Madison Cable Corp., a high volume manufacturer of electronic cable for the computer industry, and the Vice President and General Manager of the Seals Division of Ferrofluidics Corp., a major supplier to the semiconductor, disk drive and aerospace industries. Mr. Muscari began his career at Westinghouse Corporation, where he was a thermal-hydraulic engineer. Mr. Muscari holds a Bachelor of Science degree in Mechanical Engineering from Cornell University, a Master of Science degree in Mechanical Engineering from the Massachusetts Institute of Technology and a Masters of Business Administration degree from the Harvard University Graduate School of Business.
Based on Mr. Muscari’s substantial executive experience in technology-based companies and his education, the Board of Directors believes Mr. Muscari has the appropriate set of skills to serve as a member of CollabRx’s Board of Directors.
All directors hold office until our next annual meeting of the stockholders and until their successors have been duly elected or qualified. There are no family relationships between any of our directors or executive officers.
Executive Officers
The following table sets forth information regarding our executive officers as of September 1, 2014:
|
Name
|
Age
|
Position
|
|
Thomas R. Mika
|
63
|
President & CEO, Acting Chief Financial Officer, Secretary and Treasurer
|
|
Clifford Baron, Ph.D.
|
55
|
Vice President and Chief Operating Officer
|
|
Gavin Gordon, Ph.D.
|
42
|
Vice President of Business Development and Strategic Alliances
|
|
George Lundberg, M.D.
|
81
|
Editor in Chief and Chief Medical Officer
|
Thomas R. Mika’s biography is included in “Directors” above.
Clifford Baron was appointed our Vice President and Chief Operating Officer on March 5, 2014. Prior to that Dr. Baron’s most recent position was with Accelrys, Inc., a $160M+ global software provider serving pharmaceutical, biotechnology, chemical, and materials corporations. Dr. Baron has a distinguished track record leading product and go-to-market efforts in prominent life science companies. Most recently, he held the positions of Director of Biology Product Marketing, Director of Business Development and Director of Professional Services at Accelrys, Inc. He founded Lexigraphix, LLC, a strategy consultancy serving biotechnology startups with an innovative web-based system to analyze intellectual property assets. Prior to that entrepreneurial endeavor, Dr. Baron was Senior Director of Global Solutions for Applied Biosystems, Inc. (now part of Life Technologies/Thermo Fisher) where he led software product management and coordinated Applied Biosystems’ collaboration with Celera, now part of Quest Diagnostics, Inc. He was Director of Product Management at Pangea Systems / Doubletwist, a pioneering bioinformatics company, and Vice President of Product Development, Product Director and Manager of Technical Products and Information Services for Abacus Concepts, a statistical software company acquired by SAS Corp. Dr. Baron holds a BS in Biology from the University of Washington, and a Ph.D. in Biology from the University of California, Berkeley.
Gavin Gordon was appointed our Vice President of Business Development and Strategic Alliances on June 1, 2013. Prior to that Dr. Gordon served as our Head of Business Development from May 2010. Prior to joining CollabRx, Dr. Gordon was a professor at Harvard Medical School and the Co-Director of the Thoracic Surgery Oncology Laboratory at Brigham and Women’s Hospital in Boston, Massachusetts. Dr. Gordon also held an academic appointment in the Dana-Farber/Harvard Cancer Center. Dr. Gordon’s area of expertise is cancer-related therapies, medical devices and molecular diagnostics, and he has conducted technological and commercial due diligence, valuation, and financial modeling for leading healthcare, biotech, pharmaceutical, and medical devices companies. Dr. Gordon holds a B.A. degree in Chemistry from the University of North Carolina, a Ph.D. degree in Molecular and Cellular Pathology from the University of North Carolina School of Medicine, and a M.B.A. from the Franklin W. Olin Graduate School of Business at Babson College.
George Lundberg, M.D. was appointed our Chief Medical Officer on September 4, 2013. Dr. Lundberg has continued in his position as Editor-in-Chief and Chair of the Editorial Advisory Board of CollabRx. Dr. Lundberg has more than 30 years combined experience as Editor-in-Chief of JAMA (The Journal of the American Medical Association), the 10 American Medical Association specialty journals, American Medical News, Medscape, The Medscape Journal, e-Medicine from Web MD, and MedPage Today from Everyday Health. A frequent lecturer, radio and television guest, and a member of the Institute of Medicine of the National Academy of Sciences, the Industry Standard dubbed Dr. Lundberg “Online Healthcare’s Medicine Man” in 2000. Prior to joining CollabRx in 2010, Dr. Lundberg served as the President of the American Society for Clinical Pathology, Professor of Pathology and Associate Director of Laboratories at the Los Angeles County/USC Medical Center for 10 years, and for 5 years was the Professor and Chair of Pathology at the University of California, Davis. He served in the US Army during the Vietnam War, leaving as a lieutenant colonel after 11 years. Prior to his military service, Dr. Lundberg completed a clinical internship in Hawaii and a pathology residency in San Antonio. Called the medical Internet “pioneer” in 1995, Dr. Lundberg holds earned and honorary degrees from North Park College, Baylor University, the University of Alabama (Birmingham and Tuscaloosa), the State University of New York, Syracuse, Thomas Jefferson University and the Medical College of Ohio. Dr. Lundberg has worked in tropical medicine in Central America and Forensic Medicine in New York, Sweden, and England. His major professional interests are toxicology, violence, communication, physician behavior, patient safety, and health system reform. In addition to serving as Chief Medical Officer and Editor-In-Chief for CollabRx, Dr. Lundberg is presently President and Chair of the Board of Directors of The Lundberg Institute, a Consulting Professor at Stanford University, and Editor at Large for Medscape from WebMD.
There are no family relationships between any of our directors or executive officers.
Codes of Business Conduct and Ethics
Our board of directors has adopted a code of business conduct and ethics that applies to all of our employees, officers, and directors, including our Chief Executive Officer, Chief Financial Officer, and other executive and senior financial officers.
Board of Directors and Committees of the Board
Our business and affairs are managed under the direction of our Board of Directors. The number of directors is fixed by our Board of Directors, subject to the terms of our certificate of incorporation and bylaws The Board of Directors has determined each of the following current directors is an “independent director” as such term is defined in Marketplace Rule 5605(a)(2) of the Nasdaq Stock Market: Paul Billings, Jeffrey M. Krauss and Carl Muscari.
The Board of Directors has established a standing Audit Committee, a standing Compensation Committee and a standing Nominating/Corporate Governance Committee. Members will serve on these committees until their resignation or until as otherwise determined by our Board of Directors. Each of our Audit Committee, Compensation Committee and Nominating/Corporate Governance Committee is composed entirely of independent directors in accordance with current Nasdaq listing standards. Furthermore, each member of our Audit Committee meets the additional independence and financial requirements standards required by the Nasdaq Stock Market and the Securities and Exchange Commission, or SEC. The Board of Directors has further determined that Jeffrey M. Krauss, Chairman of the Audit Committee, is an “audit committee financial expert,” as such term is defined in Item 407(d)(5)(ii) of Regulation S-K promulgated by the SEC, by virtue of his relevant experience listed in his biographical summary provided above.
Audit Committee
The Audit Committee consists of Messrs. Krauss (Chairman), Billings and Muscari. The Audit Committee reviews the adequacy of internal controls and the results and scope of the audit and other services provided by the Company’s independent auditors. The Audit Committee meets periodically with management and the independent auditors. The Board of Directors has adopted an Audit Committee Charter, a copy of which is posted on our website at www.collabrx.com.
Compensation Committee
The Compensation Committee consists of Messrs. Muscari (Chairman), Billings and Krauss. The functions of the Compensation Committee include establishing salaries, incentives and other forms of compensation for our officers and other employees and administering our incentive compensation and benefit plans. The Board of Directors has adopted a compensation committee charter, a copy of which is posted on our website at www.collabrx.com.
Compensation Committee Interlocks and Insider Participation
There are no interlocking relationships between the Board of Directors or the Compensation Committee and the board of directors or compensation committee of any other company, nor has any such interlocking relationship existed in the past.
Nominating/Corporate Governance Committee
The Nominating/Corporate Governance Committee consists of Messrs. Muscari (Chairman), Krauss and Billings. The functions of the Nominating/Corporate Governance Committee are to identify qualified candidates for election to the Board of Directors and establish procedures for the director candidate nomination and evaluation. The Nominating/Corporate Governance Committee is also responsible for developing and recommending to our Board of Directors corporate governance guidelines, as well as overseeing the evaluation of our Board of Directors. The Board of Directors has adopted a Nominating/Corporate Governance Committee charter, a copy of which is posted on our website at www.collabrx.com.
Our board of directors has determined that all non-employee members of the board of directors, are independent, as determined in accordance with the rules of the NASDAQ Stock Market. In making such an independence determination, the board of directors considered the relationships that each such non-employee director has with us and all other facts and circumstances that the board of directors deemed relevant in determining their independence, including the beneficial ownership of our capital stock by each non-employee director. In considering the independence of the directors listed above, our board of directors considered the association of our directors with the holders of more than 5% of our common stock. There are no family relationships among any of our directors or executive officers.
Our board of directors may establish other committees from time to time.
None of the members of our compensation committee has at any time during the prior three years been one of our officers or employees. None of our executive officers currently serves, or in the past fiscal year has served, as a member of the board of directors or compensation committee of any entity that has one or more executive officers serving on our board of directors or compensation committee.
Limitation of Liability
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to our directors, officers, or controlling persons, we have been informed that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable.
